Impact of Microbiota: A Paradigm for Evolving Herd Immunity against Viral Diseases
- PMID: 33050511
- PMCID: PMC7599628
- DOI: 10.3390/v12101150
Impact of Microbiota: A Paradigm for Evolving Herd Immunity against Viral Diseases
Abstract
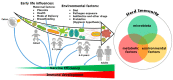
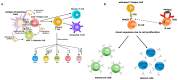
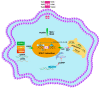
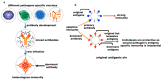
Herd immunity is the most critical and essential prophylactic intervention that delivers protection against infectious diseases at both the individual and community level. This process of natural vaccination is immensely pertinent to the current context of a pandemic caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection around the globe. The conventional idea of herd immunity is based on efficient transmission of pathogens and developing natural immunity within a population. This is entirely encouraging while fighting against any disease in pandemic circumstances. A spatial community is occupied by people having variable resistance capacity against a pathogen. Protection efficacy against once very common diseases like smallpox, poliovirus or measles has been possible only because of either natural vaccination through contagious infections or expanded immunization programs among communities. This has led to achieving herd immunity in some cohorts. The microbiome plays an essential role in developing the body's immune cells for the emerging competent vaccination process, ensuring herd immunity. Frequency of interaction among microbiota, metabolic nutrients and individual immunity preserve the degree of vaccine effectiveness against several pathogens. Microbiome symbiosis regulates pathogen transmissibility and the success of vaccination among different age groups. Imbalance of nutrients perturbs microbiota and abrogates immunity. Thus, a particular population can become vulnerable to the infection. Intestinal dysbiosis leads to environmental enteropathy (EE). As a consequence, the generation of herd immunity can either be delayed or not start in a particular cohort. Moreover, disparities of the protective response of many vaccines in developing countries outside of developed countries are due to inconsistencies of healthy microbiota among the individuals. We suggested that pan-India poliovirus vaccination program, capable of inducing herd immunity among communities for the last 30 years, may also influence the inception of natural course of heterologous immunity against SARS-CoV-2 infection. Nonetheless, this anamnestic recall is somewhat counterintuitive, as antibody generation against original antigens of SARS-CoV-2 will be subdued due to original antigenic sin.
Keywords: EE; SARS-CoV-2; herd immunity; heterologous immunity; microbiome; nutrition.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

