EULAR recommendations for a core data set for pregnancy registries in rheumatology
- PMID: 33055080
- PMCID: PMC7788063
- DOI: 10.1136/annrheumdis-2020-218356
EULAR recommendations for a core data set for pregnancy registries in rheumatology
Abstract
Background and objective: There is an urgent need for robust data on the trajectories and outcomes of pregnancies in women with inflammatory rheumatic diseases (IRD). In particular when rare outcomes or rare diseases are to be investigated, collaborative approaches are required. However, joint data analyses are often limited by the heterogeneity of the different data sources.To facilitate future research collaboration, a European League Against Rheumatism (EULAR) Task Force defined a core data set with a minimum of items to be collected by pregnancy registries in rheumatology covering the period of pregnancy and the 28-day neonatal phase in women with any underlying IRD.
Methods: A stepwise process included a two-round Delphi survey and a face-to-face meeting to achieve consensus about relevant items.
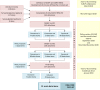
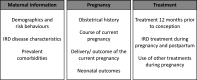
Results: A total of 64 multidisciplinary stakeholders from 14 different countries participated in the two rounds of the Delphi process. During the following face-to-face meeting of the EULAR Task Force, consensus was reached on 51 main items covering 'maternal information', 'pregnancy' and 'treatment'. Generic instruments for assessment are recommended for every item. Furthermore, for the five most frequent IRDs rheumatoid arthritis, spondyloarthritis, juvenile idiopathic arthritis, systemic lupus erythematosus and other connective tissue diseases, disease-specific laboratory markers and disease activity measurements are proposed.
Conclusion: This is the first consensus-based core data set for prospective pregnancy registries in rheumatology. Its purpose is to stimulate and facilitate multinational collaborations that aim to increase the knowledge about pregnancy course and safety of treatment in women with IRDs during pregnancy.
Keywords: antirheumatic agents; autoimmune diseases; biological therapy; epidemiology; health care; outcome assessment.
© Author(s) (or their employer(s)) 2021. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: AM: speaker/consultant fees: Abbvie, BMS, MSD, Novartis, Pfizer, UCB; unrestricted grants: Pfizer and UCBAS: speaker fees: AbbVie, BMS, MSD, Novartis, Pfizer, Roche, Sanofi Aventis, UCBA Zbinden: none. AZink: speaker fees from AbbVie, Janssen, Pfizer, Roche and Sanofi-Aventis. CN-P: consultancy fees from UCB and Alliance Pharma. Speaker fees from UCB, Alliance Pharma, Alexion, Sanofi, Falk, Janssen. DDC: none. DG: none. FF: consultant for UCB, GSK, Roche. Speakers bureau: UCB, GSK. Research grants from UCBLA: speaker/consultant fees: GSK, Novartis, UCB, Eli Lilly, INOVA Diagnostics/Werfen Group. LR: none. MW: none. NC-C: no personal fees. Research grants from UCBRD: received an unrestricted research grant form UCB Pharma BV and from the Dutch Arthritis Association a non-commercial fund raising organisation. RFB: speaker/consultant fees: Abbvie, Biogen, BMS, Celgene, Chugai, GSK, Janssen, Lilly, Medac, MSD, Novartis, Roche, UCBRÖ: None. SCR-G: speaker fees from Sanofi, MSD, UCB, Novartis and Janssen. SS: none. YM: lecture honoraria from Pfizer.
Figures