Galectin-7 downregulation in lesional keratinocytes contributes to enhanced IL-17A signaling and skin pathology in psoriasis
- PMID: 33055419
- PMCID: PMC7773376
- DOI: 10.1172/JCI130740
Galectin-7 downregulation in lesional keratinocytes contributes to enhanced IL-17A signaling and skin pathology in psoriasis
Abstract
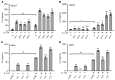
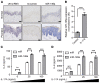
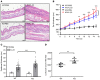
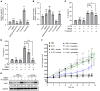
Psoriasis is a chronic inflammatory skin disease characterized by inflammatory cell infiltration, as well as hyperproliferation of keratinocytes in skin lesions, and is considered a metabolic syndrome. We found that the expression of galectin-7 is reduced in skin lesions of patients with psoriasis. IL-17A and TNF-α, 2 cytokines intimately involved in the development of psoriatic lesions, suppressed galectin-7 expression in human primary keratinocytes (HEKn cells) and the immortalized human keratinocyte cell line HaCaT. A galectin-7 knockdown in these cells elevated the production of IL-6 and IL-8 and enhanced ERK signaling when the cells were stimulated with IL-17A. Galectin-7 attenuated IL-17A-induced production of inflammatory mediators by keratinocytes via the microRNA-146a/ERK pathway. Moreover, galectin-7-deficient mice showed enhanced epidermal hyperplasia and skin inflammation in response to intradermal IL-23 injection. We identified fluvastatin as an inducer of galectin-7 expression by connectivity map analysis, confirmed this effect in keratinocytes, and demonstrated that fluvastatin attenuated IL-6 and IL-8 production induced by IL-17A. Thus, we validate a role of galectin-7 in the pathogenesis of psoriasis, in both epidermal hyperplasia and keratinocyte-mediated inflammatory responses, and formulate a rationale for the use of statins in the treatment of psoriasis.
Keywords: Autoimmune diseases; Autoimmunity; Dermatology; Skin.
Conflict of interest statement
Figures



Similar articles
-
Galectin-8 Is Upregulated in Keratinocytes by IL-17A and Promotes Proliferation by Regulating Mitosis in Psoriasis.J Invest Dermatol. 2021 Mar;141(3):503-511.e9. doi: 10.1016/j.jid.2020.07.021. Epub 2020 Aug 15. J Invest Dermatol. 2021. PMID: 32805218
-
CD8(+) T cells mediate RAS-induced psoriasis-like skin inflammation through IFN-γ.J Invest Dermatol. 2013 Apr;133(4):955-63. doi: 10.1038/jid.2012.390. Epub 2012 Nov 15. J Invest Dermatol. 2013. PMID: 23151849 Free PMC article.
-
Involvement of IL-17F via the induction of IL-6 in psoriasis.Arch Dermatol Res. 2010 Sep;302(7):499-505. doi: 10.1007/s00403-010-1033-8. Epub 2010 Feb 11. Arch Dermatol Res. 2010. PMID: 20148256
-
Interleukin-17A and Keratinocytes in Psoriasis.Int J Mol Sci. 2020 Feb 13;21(4):1275. doi: 10.3390/ijms21041275. Int J Mol Sci. 2020. PMID: 32070069 Free PMC article. Review.
-
Pathophysiology of psoriasis: recent advances on IL-23 and Th17 cytokines.Curr Rheumatol Rep. 2007 Dec;9(6):461-7. doi: 10.1007/s11926-007-0075-1. Curr Rheumatol Rep. 2007. PMID: 18177599 Free PMC article. Review.
Cited by
-
Histone and Histone Acetylation-Related Alterations of Gene Expression in Uninvolved Psoriatic Skin and Their Effects on Cell Proliferation, Differentiation, and Immune Responses.Int J Mol Sci. 2023 Sep 26;24(19):14551. doi: 10.3390/ijms241914551. Int J Mol Sci. 2023. PMID: 37833997 Free PMC article.
-
Immunosuppression causes dynamic changes in expression QTLs in psoriatic skin.Nat Commun. 2023 Oct 7;14(1):6268. doi: 10.1038/s41467-023-41984-2. Nat Commun. 2023. PMID: 37805522 Free PMC article.
-
PIN1‑silencing mitigates keratinocyte proliferation and the inflammatory response in psoriasis by activating mitochondrial autophagy.Exp Ther Med. 2024 Aug 19;28(4):402. doi: 10.3892/etm.2024.12691. eCollection 2024 Oct. Exp Ther Med. 2024. PMID: 39234585 Free PMC article.
-
Medawar's PostEra: Galectins Emerged as Key Players During Fetal-Maternal Glycoimmune Adaptation.Front Immunol. 2021 Dec 15;12:784473. doi: 10.3389/fimmu.2021.784473. eCollection 2021. Front Immunol. 2021. PMID: 34975875 Free PMC article. Review.
-
Dupilumab-associated head and neck dermatitis shows a pronounced type 22 immune signature mediated by oligoclonally expanded T cells.Nat Commun. 2024 Apr 2;15(1):2839. doi: 10.1038/s41467-024-46540-0. Nat Commun. 2024. PMID: 38565563 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

