PIEZO2 in sensory neurons and urothelial cells coordinates urination
- PMID: 33057202
- PMCID: PMC7725878
- DOI: 10.1038/s41586-020-2830-7
PIEZO2 in sensory neurons and urothelial cells coordinates urination
Abstract
Henry Miller stated that "to relieve a full bladder is one of the great human joys". Urination is critically important in health and ailments of the lower urinary tract cause high pathological burden. Although there have been advances in understanding the central circuitry in the brain that facilitates urination1-3, there is a lack of in-depth mechanistic insight into the process. In addition to central control, micturition reflexes that govern urination are all initiated by peripheral mechanical stimuli such as bladder stretch and urethral flow4. The mechanotransduction molecules and cell types that function as the primary stretch and pressure detectors in the urinary tract mostly remain unknown. Here we identify expression of the mechanosensitive ion channel PIEZO2 in lower urinary tract tissues, where it is required for low-threshold bladder-stretch sensing and urethral micturition reflexes. We show that PIEZO2 acts as a sensor in both the bladder urothelium and innervating sensory neurons. Humans and mice lacking functional PIEZO2 have impaired bladder control, and humans lacking functional PIEZO2 report deficient bladder-filling sensation. This study identifies PIEZO2 as a key mechanosensor in urinary function. These findings set the foundation for future work to identify the interactions between urothelial cells and sensory neurons that control urination.
Conflict of interest statement
The authors declare no competing interests.
Figures
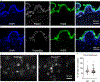
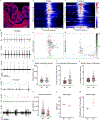
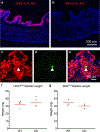

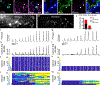


Comment in
-
PIEZO2 in urinary function.Nat Rev Urol. 2020 Dec;17(12):658. doi: 10.1038/s41585-020-00395-4. Nat Rev Urol. 2020. PMID: 33173207 No abstract available.
-
PIEZO2, a mechanosensor in the urinary bladder.Kidney Int. 2021 Jul;100(1):9-11. doi: 10.1016/j.kint.2021.02.021. Epub 2021 Mar 2. Kidney Int. 2021. PMID: 33667505 No abstract available.
-
Re: PIEZO2 in Sensory Neurons and Urothelial Cells Coordinate Urination.Eur Urol. 2021 Aug;80(2):255-256. doi: 10.1016/j.eururo.2021.04.002. Epub 2021 Apr 17. Eur Urol. 2021. PMID: 33875308 No abstract available.
References
-
- Garry RC, Roberts TD & Todd JK Reflex responses of the external urethral sphincter of the cat to filling of the bladder. J Physiol 139, 13–14P (1957). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

