SynBio-SynChem Approaches to Diversifying the Pacidamycins through the Exploitation of an Observed Pictet-Spengler Reaction
- PMID: 33058439
- PMCID: PMC7898326
- DOI: 10.1002/cbic.202000594
SynBio-SynChem Approaches to Diversifying the Pacidamycins through the Exploitation of an Observed Pictet-Spengler Reaction
Abstract
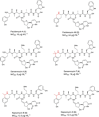
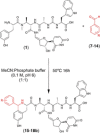
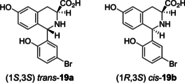
A nonenzymatic Pictet-Spengler reaction has been postulated to give rise to a subset of naturally occurring uridyl peptide antibiotics (UPAs). Here, using a combination of strain engineering and synthetic chemistry, we demonstrate that Pictet-Spengler chemistry may be employed to generate even greater diversity in the UPAs. We use an engineered strain to afford access to meta-tyrosine containing pacidamycin 4. Pictet-Spengler diversification of this compound using a small series of aryl-aldehydes was achieved with some derivatives affording remarkable diastereomeric control.
Keywords: Pictet-Spengler; compound diversification; natural products; pacidamycin; semisynthesis; uridyl peptide antibiotic.
© 2020 The Authors. ChemBioChem published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Karwowski J. P., Jackson M., Theriault R. J., Chen R. H., Barlow G. J., Maus M. L., J. Antibiot. 1989, 42, 506–511. - PubMed
-
- Chatterjee S., Nadkarni S. R., Vijayakumar E. K., Patel M. V., Gangul B. N., Fehlhaber H. W., Vertesy L., J. Antibiot. 1994, 47, 595–598. - PubMed
-
- Xie Y., Chen R., Si S., Sun C., Xu H., J. Antibiot. 2007, 60, 158–161. - PubMed
-
- Xie Y., Xu H., Si S., Sun C., Chen R., J. Antibiot. 2008, 61, 237–240. - PubMed
-
- Inukai M., Isono F., Takahashi S., Enokita R., Sakaida Y., Haneishi T., J. Antibiot. 1989, 42, 662–666. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

