RNA-based thermoregulation of a Campylobacter jejuni zinc resistance determinant
- PMID: 33064782
- PMCID: PMC7592916
- DOI: 10.1371/journal.ppat.1009008
RNA-based thermoregulation of a Campylobacter jejuni zinc resistance determinant
Abstract
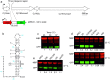
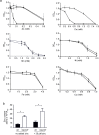
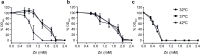
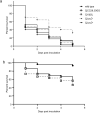
RNA thermometers (RNATs) trigger bacterial virulence factor expression in response to the temperature shift on entering a warm-blooded host. At lower temperatures these secondary structures sequester ribosome-binding sites (RBSs) to prevent translation initiation, whereas at elevated temperatures they "melt" allowing translation. Campylobacter jejuni is the leading bacterial cause of human gastroenteritis worldwide yet little is known about how it interacts with the host including host induced gene regulation. Here we demonstrate that an RNAT regulates a C. jejuni gene, Cj1163c or czcD, encoding a member of the Cation Diffusion Facilitator family. The czcD upstream untranslated region contains a predicted stem loop within the mRNA that sequesters the RBS to inhibit translation at temperatures below 37°C. Mutations that disrupt or enhance predicted secondary structure have significant and predictable effects on temperature regulation. We also show that in an RNAT independent manner, CzcD expression is induced by Zn(II). Mutants lacking czcD are hypersensitive to Zn(II) and also over-accumulate Zn(II) relative to wild-type, all consistent with CzcD functioning as a Zn(II) exporter. Importantly, we demonstrate that C. jejuni Zn(II)-tolerance at 32°C, a temperature at which the RNAT limits CzcD production, is increased by RNAT disruption. Finally we show that czcD inactivation attenuates larval killing in a Galleria infection model and that at 32°C disrupting RNAT secondary structure to allow CzcD production can enhance killing. We hypothesise that CzcD regulation by metals and temperature provides a mechanism for C. jejuni to overcome innate immune system-mediated Zn(II) toxicity in warm-blooded animal hosts.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Kirk MD, Pires SM, Black RE, Caipo M, Crump JA, Devleesschauwer B, et al. World Health Organization Estimates of the Global and Regional Disease Burden of 22 Foodborne Bacterial, Protozoal, and Viral Diseases, 2010: A Data Synthesis. PLoS Med. 2015;12(12):e1001921 10.1371/journal.pmed.1001921 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

