Activation of the VEGF-A/ERK/PLA2 Axis Mediates Early Retinal Endothelial Cell Damage Induced by High Glucose: New Insight from an In Vitro Model of Diabetic Retinopathy
- PMID: 33065984
- PMCID: PMC7589177
- DOI: 10.3390/ijms21207528
Activation of the VEGF-A/ERK/PLA2 Axis Mediates Early Retinal Endothelial Cell Damage Induced by High Glucose: New Insight from an In Vitro Model of Diabetic Retinopathy
Abstract
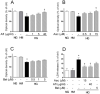
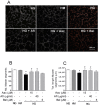
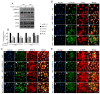
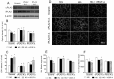
Early blood retinal barrier (BRB) dysfunction induced by hyperglycemia was related to increased pro-inflammatory activity of phospholipase A2 (PLA2) and the upregulation of vascular endothelial growth factor A (VEGF-A). Here, we tested the role of VEGF-A in high glucose (HG)-induced damage of human retinal endothelial cells (HRECs) mediated by Ca++-dependent (cPLA2) and Ca++-independent (iPLA2) PLA2s. HRECs were treated with normal glucose (5 mM, NG) or high glucose (25 mM, HG) for 48 h with or without the VEGF-trap Aflibercept (Afl, 40 µg/mL), the cPLA2 inhibitor arachidonoyl trifluoromethyl ketone (AACOCF3; 15 µM), the iPLA2 inhibitor bromoenol lactone (BEL; 5 µM), or VEGF-A (80 ng/mL). Both Afl and AACOCF3 prevented HG-induced damage (MTT and LDH release), impairment of angiogenic potential (tube-formation), and expression of VEGF-A mRNA. Furthermore, Afl counteracted HG-induced increase of phospho-ERK and phospho-cPLA2 (immunoblot). VEGF-A in HG-medium increased glucose toxicity, through upregulation of phospho-ERK, phospho-cPLA2, and iPLA2 (about 55%, 45%, and 50%, respectively); immunocytochemistry confirmed the activation of these proteins. cPLA2 knockdown by siRNA entirely prevented cell damage induced by HG or by HG plus VEGF-A, while iPLA2 knockdown produced a milder protective effect. These data indicate that VEGF-A mediates the early glucose-induced damage in retinal endothelium through the involvement of ERK1/2/PLA2 axis activation.
Keywords: Aflibercept; VEGF-A; high glucose; phospholipase A2; retinal endothelium.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Aflibercept, bevacizumab and ranibizumab prevent glucose-induced damage in human retinal pericytes in vitro, through a PLA2/COX-2/VEGF-A pathway.Biochem Pharmacol. 2015 Aug 1;96(3):278-87. doi: 10.1016/j.bcp.2015.05.017. Epub 2015 Jun 6. Biochem Pharmacol. 2015. PMID: 26056075
-
Sulodexide prevents activation of the PLA2/COX-2/VEGF inflammatory pathway in human retinal endothelial cells by blocking the effect of AGE/RAGE.Biochem Pharmacol. 2017 Oct 15;142:145-154. doi: 10.1016/j.bcp.2017.06.130. Epub 2017 Jun 23. Biochem Pharmacol. 2017. PMID: 28651842
-
Role of phospholipases A2 in diabetic retinopathy: in vitro and in vivo studies.Biochem Pharmacol. 2013 Dec 1;86(11):1603-13. doi: 10.1016/j.bcp.2013.09.008. Epub 2013 Sep 25. Biochem Pharmacol. 2013. PMID: 24076420
-
Phospholipase A2 Isoforms as Novel Targets for Prevention and Treatment of Inflammatory and Oncologic Diseases.Curr Drug Targets. 2016;17(16):1940-1962. doi: 10.2174/1389450116666150727122501. Curr Drug Targets. 2016. PMID: 26212262 Review.
-
Small-molecule inhibitors as potential therapeutics and as tools to understand the role of phospholipases A2.Biochim Biophys Acta Mol Cell Biol Lipids. 2019 Jun;1864(6):941-956. doi: 10.1016/j.bbalip.2018.08.009. Epub 2018 Aug 23. Biochim Biophys Acta Mol Cell Biol Lipids. 2019. PMID: 30905350 Free PMC article. Review.
Cited by
-
The palliative effects of folic acid on retinal microvessels in diabetic retinopathy via regulating the metabolism of DNA methylation and hydroxymethylation.Bioengineered. 2021 Dec;12(2):10766-10774. doi: 10.1080/21655979.2021.2003924. Bioengineered. 2021. PMID: 34874218 Free PMC article.
-
YTHDF3-mediated FLCN/cPLA2 axis improves cardiac fibrosis via suppressing lysosomal function.Acta Pharmacol Sin. 2025 May;46(5):1262-1274. doi: 10.1038/s41401-024-01425-2. Epub 2025 Jan 13. Acta Pharmacol Sin. 2025. PMID: 39806064
-
Randomised, multicentre, placebo-controlled trial of fenofibrate for treatment of diabetic macular oedema with economic evaluation (FORTE study): study protocol for a randomised control trial.BMJ Open. 2024 Dec 20;14(12):e089518. doi: 10.1136/bmjopen-2024-089518. BMJ Open. 2024. PMID: 39806595 Free PMC article.
-
Effects of High Glucose Concentration on Pericyte-Like Differentiated Human Adipose-Derived Mesenchymal Stem Cells.Int J Mol Sci. 2021 Apr 27;22(9):4604. doi: 10.3390/ijms22094604. Int J Mol Sci. 2021. PMID: 33925714 Free PMC article.
-
Tetramethylpyrazine Ameliorates Peritoneal Angiogenesis by Regulating VEGF/Hippo/YAP Signaling.Front Pharmacol. 2021 Apr 13;12:649581. doi: 10.3389/fphar.2021.649581. eCollection 2021. Front Pharmacol. 2021. PMID: 33927624 Free PMC article.
References
-
- Ma¨kimattila S., Virkama¨ki A., Groop P.-H., Cockcroft J., Utriainen T., Fagerudd J., Yki-Ja¨rvinen H. Chronic Hyperglycemia Impairs Endothelial Function and Insulin Sensitivity Via Different Mechanisms in Insulin-Dependent Diabetes Mellitus. Circulation. 1996;94:1276–1282. doi: 10.1161/01.CIR.94.6.1276. - DOI - PubMed
-
- Chou J., Rollins S., Fawzi A.A. Single Molecule and Single Cell Sequencing. Volume 801. Springer Science and Business Media LLC; Berlin/Heidelberg, Germany: 2014. Role of endothelial cell and pericyte dysfunction in diabetic retinopathy: Review of techniques in rodent models; pp. 669–675. - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

