Cytotoxicity and Mitochondrial Dysregulation Caused by α-Synuclein in Dictyostelium discoideum
- PMID: 33066427
- PMCID: PMC7602147
- DOI: 10.3390/cells9102289
Cytotoxicity and Mitochondrial Dysregulation Caused by α-Synuclein in Dictyostelium discoideum
Abstract
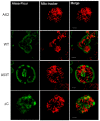
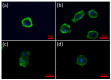
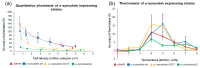
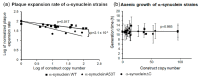
Alpha synuclein has been linked to both sporadic and familial forms of Parkinson's disease (PD) and is the most abundant protein in Lewy bodies a hallmark of Parkinson's disease. The function of this protein and the molecular mechanisms underlying its toxicity are still unclear, but many studies have suggested that the mechanism of α-synuclein toxicity involves alterations to mitochondrial function. Here we expressed human α-synuclein and two PD-causing α-synuclein mutant proteins (with a point mutation, A53T, and a C-terminal 20 amino acid truncation) in the eukaryotic model Dictyostelium discoideum. Mitochondrial disease has been well studied in D. discoideum and, unlike in mammals, mitochondrial dysfunction results in a clear set of defective phenotypes. These defective phenotypes are caused by the chronic hyperactivation of the cellular energy sensor, AMP-activated protein kinase (AMPK). Expression of α-synuclein wild type and mutant forms was toxic to the cells and mitochondrial function was dysregulated. Some but not all of the defective phenotypes could be rescued by down regulation of AMPK revealing both AMPK-dependent and -independent mechanisms. Importantly, we also show that the C-terminus of α-synuclein is required and sufficient for the localisation of the protein to the cell cortex in D. discoideum.
Keywords: AMPK; Dictyostelium; mitochondria; α-synuclein.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Flagmeier P., Meisl G., Vendruscolo M., Knowles T.P., Dobson C.M., Buell A.K., Galvagnion C. Mutations associated with familial Parkinson’s disease alter the initiation and amplification steps of alpha-synuclein aggregation. Proc. Natl. Acad. Sci. USA. 2016;113:10328–10333. doi: 10.1073/pnas.1604645113. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

