Inter- vs. Intramolecular Hydrogen Bond Patterns and Proton Dynamics in Nitrophthalic Acid Associates
- PMID: 33066679
- PMCID: PMC7587347
- DOI: 10.3390/molecules25204720
Inter- vs. Intramolecular Hydrogen Bond Patterns and Proton Dynamics in Nitrophthalic Acid Associates
Abstract
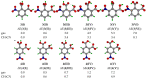
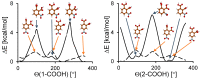
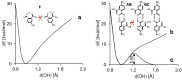
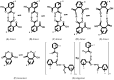
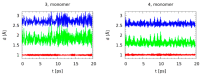
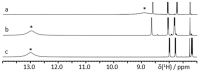
Noncovalent interactions are among the main tools of molecular engineering. Rational molecular design requires knowledge about a result of interplay between given structural moieties within a given phase state. We herein report a study of intra- and intermolecular interactions of 3-nitrophthalic and 4-nitrophthalic acids in the gas, liquid, and solid phases. A combination of the Infrared, Raman, Nuclear Magnetic Resonance, and Incoherent Inelastic Neutron Scattering spectroscopies and the Car-Parrinello Molecular Dynamics and Density Functional Theory calculations was used. This integrated approach made it possible to assess the balance of repulsive and attractive intramolecular interactions between adjacent carboxyl groups as well as to study the dependence of this balance on steric confinement and the effect of this balance on intermolecular interactions of the carboxyl groups.
Keywords: CPMD; DFT; IINS; IR; NMR; Raman; carboxyl group; proton dynamics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
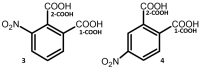
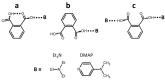
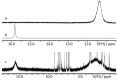
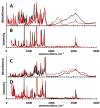
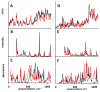
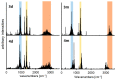
References
-
- Shenderovich I.G., Limbach H.-H., Smirnov S.N., Tolstoy P.M., Denisov G.S., Golubev N.S. H/D isotope effects on the low-temperature NMR parameters and hydrogen bond geometries of (FH)2F− and (FH)3F− dissolved in CDF3/CDF2Cl. Phys. Chem. Chem. Phys. 2002;4:5488–5497. doi: 10.1039/B206323A. - DOI
-
- Mauder D., Akcakayiran D., Lesnichin S.B., Findenegg G.H., Shenderovich I.G. Acidity of Sulfonic and Phosphonic Acid-Functionalized SBA-15 under Almost Water-Free Conditions. J. Phys. Chem. C. 2009;113:19185–19192. doi: 10.1021/jp907058y. - DOI
-
- Melikova S.M., Voronin A.P., Panek J., Frolov N.E., Shishkina A.V., Rykounov A.A., Tretyakov P.Y., Vener M.V. Interplay of pi-stacking and inter-stacking interactions in two-component crystals of neutral closed-shell aromatic compounds: Periodic DFT study. RSC Adv. 2020;10:27899–27910. doi: 10.1039/D0RA04799F. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

