Two Type VI Secretion Systems of Enterobacter cloacae Are Required for Bacterial Competition, Cell Adherence, and Intestinal Colonization
- PMID: 33072020
- PMCID: PMC7541819
- DOI: 10.3389/fmicb.2020.560488
Two Type VI Secretion Systems of Enterobacter cloacae Are Required for Bacterial Competition, Cell Adherence, and Intestinal Colonization
Abstract
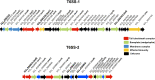
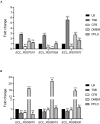
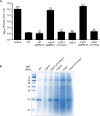
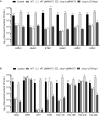
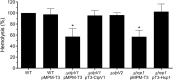
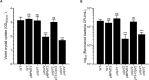
Enterobacter cloacae has emerged as an opportunistic pathogen in healthcare-associated infections. Analysis of the genomic sequences of several E. cloacae strains revealed the presence of genes that code for expression of at least one type VI secretion system (T6SS). Here, we report that E. cloacae strain ATCC 13047 codes for two functional T6SS named T6SS-1 and T6SS-2. T6SS-1 and T6SS-2 were preferentially expressed in tryptic soy broth and tissue culture medium (DMEM), respectively. Mutants in T6SS-1-associated genes clpV1 and hcp1 significantly affected their ability of inter- and intra-bacterial killing indicating that T6SS-1 is required for bacterial competition. In addition, the Hcp effector protein was detected in supernatants of E. cloacae cultures and a functional T6SS-1 was required for the secretion of this protein. A clpV2 mutant was impaired in both biofilm formation and adherence to epithelial cells, supporting the notion that these phenotypes are T6SS-2 dependent. In vivo data strongly suggest that both T6SSs are required for intestinal colonization because single and double mutants in clpV1 and clpV2 genes were defective in gut colonization in mice. We conclude that the two T6SSs are involved in the pathogenesis scheme of E. cloacae with specialized functions in the interaction with other bacteria and with host cells.
Keywords: ClpV; E. cloacae; Hcp; T6SS; virulence.
Copyright © 2020 Soria-Bustos, Ares, Gómez-Aldapa, González-y-Merchand, Girón and De la Cruz.
Figures

References
-
- Ares M. A., Abundes-Gallegos J., Rodriguez-Valverde D., Panunzi L. G., Jimenez-Galicia C., Jarillo-Quijada M. D., et al. (2019). The Coli Surface Antigen CS3 of Enterotoxigenic Escherichia coli is differentially regulated by H-NS, CRP, and CpxRA Global Regulators. Front. Microbiol. 10:1685. 10.3389/fmicb.2019.01685 - DOI - PMC - PubMed
-
- Ares M. A., Fernandez-Vazquez J. L., Rosales-Reyes R., Jarillo-Quijada M. D., Von Bargen K., Torres J., et al. (2016). H-NS Nucleoid Protein controls virulence features of Klebsiella pneumoniae by regulating the expression of type 3 Pili and the capsule polysaccharide. Front. Cell. Infect. Microbiol. 6:13. 10.3389/fcimb.2016.00013 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

