CNS Macrophages and Infant Infections
- PMID: 33072074
- PMCID: PMC7531029
- DOI: 10.3389/fimmu.2020.02123
CNS Macrophages and Infant Infections
Abstract
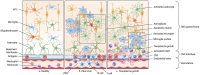
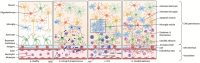
The central nervous system (CNS) harbors its own immune system composed of microglia in the parenchyma and CNS-associated macrophages (CAMs) in the perivascular space, leptomeninges, dura mater, and choroid plexus. Recent advances in understanding the CNS resident immune cells gave new insights into development, maturation and function of its immune guard. Microglia and CAMs undergo essential steps of differentiation and maturation triggered by environmental factors as well as intrinsic transcriptional programs throughout embryonic and postnatal development. These shaping steps allow the macrophages to adapt to their specific physiological function as first line of defense of the CNS and its interfaces. During infancy, the CNS might be targeted by a plethora of different pathogens which can cause severe tissue damage with potentially long reaching defects. Therefore, an efficient immune response of infant CNS macrophages is required even at these early stages to clear the infections but may also lead to detrimental consequences for the developing CNS. Here, we highlight the recent knowledge of the infant CNS immune system during embryonic and postnatal infections and the consequences for the developing CNS.
Keywords: CNS-associated macrophages; TORCH; maternal immune activation; microglia; postnatal infections; prenatal infections.
Copyright © 2020 Oschwald, Petry, Kierdorf and Erny.
Figures

Similar articles
-
Macrophages at CNS interfaces: ontogeny and function in health and disease.Nat Rev Neurosci. 2019 Sep;20(9):547-562. doi: 10.1038/s41583-019-0201-x. Epub 2019 Jul 29. Nat Rev Neurosci. 2019. PMID: 31358892 Review.
-
A comprehensive review: neuroinflammation and immune communication between the central nervous system and the periphery.Cytokine. 2025 Aug;192:156974. doi: 10.1016/j.cyto.2025.156974. Epub 2025 May 30. Cytokine. 2025. PMID: 40449035 Review.
-
Contributions of maternal and fetal antiviral immunity in congenital disease.Science. 2020 May 8;368(6491):608-612. doi: 10.1126/science.aaz1960. Science. 2020. PMID: 32381717 Review.
-
The niche matters: origin, function and fate of CNS-associated macrophages during health and disease.Acta Neuropathol. 2024 Feb 12;147(1):37. doi: 10.1007/s00401-023-02676-9. Acta Neuropathol. 2024. PMID: 38347231 Free PMC article. Review.
-
How the gut microbiota impacts neurodegenerative diseases by modulating CNS immune cells.J Neuroinflammation. 2025 Mar 3;22(1):60. doi: 10.1186/s12974-025-03371-0. J Neuroinflammation. 2025. PMID: 40033338 Free PMC article. Review.
Cited by
-
Perinatal development of innate immune topology.Elife. 2021 May 25;10:e67793. doi: 10.7554/eLife.67793. Elife. 2021. PMID: 34032570 Free PMC article. Review.
-
The microbiota-microglia axis in central nervous system disorders.Brain Pathol. 2020 Nov;30(6):1159-1177. doi: 10.1111/bpa.12908. Epub 2020 Nov 5. Brain Pathol. 2020. PMID: 33073887 Free PMC article. Review.
-
Interactions between CNS and immune cells in tuberculous meningitis.Front Immunol. 2024 Feb 1;15:1326859. doi: 10.3389/fimmu.2024.1326859. eCollection 2024. Front Immunol. 2024. PMID: 38361935 Free PMC article. Review.
-
Knowledge gaps in Alzheimer's disease immune biomarker research.Alzheimers Dement. 2021 Dec;17(12):2030-2042. doi: 10.1002/alz.12342. Epub 2021 May 13. Alzheimers Dement. 2021. PMID: 33984178 Free PMC article. Review.
-
Immunometabolic Modulatory Role of Naltrexone in BV-2 Microglia Cells.Int J Mol Sci. 2021 Aug 5;22(16):8429. doi: 10.3390/ijms22168429. Int J Mol Sci. 2021. PMID: 34445130 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

