Pancreatitis-Associated Protein in Neonatal Screening for Cystic Fibrosis: Strengths and Weaknesses
- PMID: 33073025
- PMCID: PMC7422993
- DOI: 10.3390/ijns6020028
Pancreatitis-Associated Protein in Neonatal Screening for Cystic Fibrosis: Strengths and Weaknesses
Abstract
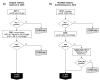
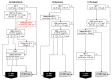
There are currently four countries and one local region in Europe that use PAP in their newborn screening programme. The first country to employ PAP at a national level was the Netherlands, which started using IRT/PAP/DNA/EGA in 2011. Germany followed in 2016 with a slightly different IRT/PAP/DNA strategy. Portugal also started in 2016, but with an IRT/PAP/IRT programme, and in 2017, Austria changed its IRT/IRT protocol to an IRT/PAP/IRT program. In 2018, Catalonia started to use an IRT/PAP/IRT/DNA strategy. The strengths of PAP are the avoidance of carrier detection and a lower detection rate of CFSPID. PAP seems to have advantages in detecting CF in ethnically-diverse populations, as it is a biochemical approach to screening, which looks for pancreatic injury. Compared to an IRT/IRT protocol, an IRT/PAP protocol leads to earlier diagnoses. While PAP can be assessed with the same screening card as the first IRT, the second IRT in an IRT/IRT protocol requires a second heel prick around the 21st day of the patient's life. However, IRT/PAP has two main weaknesses. First, an IRT/PAP protocol seems to have a lower sensitivity compared to a well-functioning IRT/DNA protocol, and second, IRT/PAP that is performed as a purely biochemical protocol has a very low positive predictive value. However, if the advantages of PAP are to be exploited, a combination of IRT/PAP with genetic screening or a second IRT as a third tier could be an alternative for a sufficiently performing CF-NBS protocol.
Keywords: biochemical screening; cystic fibrosis; immunoreactive trypsinogen; newborn screening; pancreatitis associated protein.
© 2020 by the authors.
Conflict of interest statement
Conflicts of InterestThe authors declare no conflict of interest.
Figures
Similar articles
-
Initial evaluation of a biochemical cystic fibrosis newborn screening by sequential analysis of immunoreactive trypsinogen and pancreatitis-associated protein (IRT/PAP) as a strategy that does not involve DNA testing in a Northern European population.J Inherit Metab Dis. 2010 Oct;33(Suppl 2):S263-71. doi: 10.1007/s10545-010-9174-7. Epub 2010 Aug 17. J Inherit Metab Dis. 2010. PMID: 20714932
-
Five years of experience with biochemical cystic fibrosis newborn screening based on IRT/PAP in Germany.Pediatr Pulmonol. 2015 Jul;50(7):655-64. doi: 10.1002/ppul.23190. Epub 2015 Apr 23. Pediatr Pulmonol. 2015. PMID: 25914230
-
Final results of the southwest German pilot study on cystic fibrosis newborn screening - Evaluation of an IRT/PAP protocol with IRT-dependent safety net.J Cyst Fibros. 2022 May;21(3):422-433. doi: 10.1016/j.jcf.2021.10.007. Epub 2021 Nov 9. J Cyst Fibros. 2022. PMID: 34764021
-
Severe lung disease in children with cystic fibrosis missed in newborn screening.Pediatr Pulmonol. 2024 Jan;59(1):163-168. doi: 10.1002/ppul.26734. Epub 2023 Oct 27. Pediatr Pulmonol. 2024. PMID: 37888495 Review.
-
History of Newborn Screening for Cystic Fibrosis-The Early Years.Int J Neonatal Screen. 2020 Jan 31;6(1):8. doi: 10.3390/ijns6010008. eCollection 2020 Mar. Int J Neonatal Screen. 2020. PMID: 33073006 Free PMC article. Review.
Cited by
-
Four-year evaluation of neonatal cystic fibrosis screening in Southern Belgium.Eur J Pediatr. 2024 Nov 21;184(1):38. doi: 10.1007/s00431-024-05845-4. Eur J Pediatr. 2024. PMID: 39570414
-
Newborn Screening Program for Cystic Fibrosis in Türkiye: Experiences from False-Negative Tests and Requirement for Optimization.Balkan Med J. 2025 Jan 2;42(1):45-53. doi: 10.4274/balkanmedj.galenos.2024.2024-7-144. Epub 2024 Nov 22. Balkan Med J. 2025. PMID: 39574132 Free PMC article.
-
IRT/IRT as a newborn cystic fibrosis screening method: optimal cutoff points for a mixed population.Cad Saude Publica. 2024 Aug 26;40(7):e00150623. doi: 10.1590/0102-311XEN150623. eCollection 2024. Cad Saude Publica. 2024. PMID: 39194088 Free PMC article.
-
Neonatal Screening for Cystic Fibrosis in Hungary-First-Year Experiences.Int J Neonatal Screen. 2023 Aug 23;9(3):47. doi: 10.3390/ijns9030047. Int J Neonatal Screen. 2023. PMID: 37754773 Free PMC article.
-
Current Status of Newborn Bloodspot Screening Worldwide 2024: A Comprehensive Review of Recent Activities (2020-2023).Int J Neonatal Screen. 2024 May 23;10(2):38. doi: 10.3390/ijns10020038. Int J Neonatal Screen. 2024. PMID: 38920845 Free PMC article. Review.
References
-
- Iovanna J.L., Férec C., Sarles J., Dagorn J.C. The pancreatitis-associated protein (PAP). A new candidate for neonatal screening of cystic fibrosis. Comptes Rendus de l’Académie des Sciences Series III Sciences de la Vie. 1994;317:561–564. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

