Multiple Reaction Monitoring Mass Spectrometry for the Drug Monitoring of Ivacaftor, Tezacaftor, and Elexacaftor Treatment Response in Cystic Fibrosis: A High-Throughput Method
- PMID: 33073196
- PMCID: PMC7551731
- DOI: 10.1021/acsptsci.0c00103
Multiple Reaction Monitoring Mass Spectrometry for the Drug Monitoring of Ivacaftor, Tezacaftor, and Elexacaftor Treatment Response in Cystic Fibrosis: A High-Throughput Method
Abstract
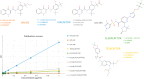
Ivacaftor-tezacaftor and ivacaftor-tezacaftor-elexacaftor are new breakthrough cystic fibrosis (CF) drug combinations that directly modulate the activity and trafficking of the defective CF transmembrane conductance regulator protein (CFTR) underlying the CF disease state. Currently, in the hospital setting, there are no therapeutic drug monitoring assays for these very expensive, albeit, life-saving drugs. A rapid and precise novel method for the quantification of ivacaftor, its metabolites, tezacaftor, and elexacaftor, in human plasma was developed and validated using multiple reaction monitoring mass spectrometry (MRM/MS). The MRM/MS analytical method was validated at a concentration range of 0.0025-1 μg/mL for ivacaftor, ivacaftor-M1, ivacaftor-M6, tezacaftor, and elexacaftor in human plasma. The method displayed good accuracy (90.62-94.51%) and reproducibility (99.91-100%) including at low concentrations 0.01 μg/mL. With a mobile phase consisting of [acetonitrile/water]/0.1% formic acid (70:30 v/v) at a flow rate of 0.5 mL/min, a linear correlation was observed over a concentration range of 0.0025-1 μg/mL in human plasma for ivacaftor (R 2 = 0.9865105), ivacaftor-M1 (R 2 = 0.9852684), ivacaftor-M6 (R 2 = 0.9911764), tezacaftor (R 2 = 0.98742470), and elexacaftor (R 2 = 0.9897608). The reported method can accurately quantify ivacaftor, ivacaftor-M1, ivacaftor-M6, tezacaftor, and elexacaftor at low concentrations in human plasma. We have established a cost-efficient and timely method for measuring ivacaftor, its metabolites, and tezacaftor with or without elexacaftor in human plasma suitable for high-throughput applications in the hospital settings or clinical trials.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
References
-
- Taylor-Cousar J. L.; Munck A.; McKone E. F.; van der Ent C. K.; Moeller A.; Simard C.; Wang L. T.; Ingenito E. P.; McKee C.; Lu Y.; Lekstrom-Himes J.; Elborn J. S. (2017) Tezacaftor-Ivacaftor in Patients with Cystic Fibrosis Homozygous for Phe508del. N. Engl. J. Med. 377 (21), 2013–2023. 10.1056/NEJMoa1709846. - DOI - PubMed
-
- Rowe S. M.; McColley S. A.; Rietschel E.; Li X.; Bell S. C.; Konstan M. W.; Marigowda G.; Waltz D.; Boyle M. P.; Group V. X. S. (2017) Lumacaftor/Ivacaftor Treatment of Patients with Cystic Fibrosis Heterozygous for F508del-CFTR. Ann. Am. Thorac Soc. 14 (2), 213–219. 10.1513/AnnalsATS.201609-689OC. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases