Advancing the Logic of Chemical Synthesis: C-H Activation as Strategic and Tactical Disconnections for C-C Bond Construction
- PMID: 33073459
- PMCID: PMC8177825
- DOI: 10.1002/anie.202011901
Advancing the Logic of Chemical Synthesis: C-H Activation as Strategic and Tactical Disconnections for C-C Bond Construction
Abstract
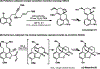
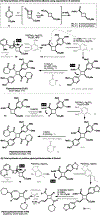
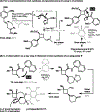
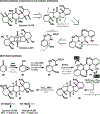
The design of synthetic routes by retrosynthetic logic is decisively influenced by the transformations available. Transition-metal-catalyzed C-H activation has emerged as a powerful strategy for C-C bond formation, with myriad methods developed for diverse substrates and coupling partners. However, its uptake in total synthesis has been tepid, partially due to their apparent synthetic intractability, as well as a lack of comprehensive guidelines for implementation. This Review addresses these issues and offers a guide to identify retrosynthetic opportunities to generate C-C bonds by C-H activation processes. By comparing total syntheses accomplished using traditional approaches and recent C-H activation methods, this Review demonstrates how C-H activation enabled C-C bond construction has led to more efficient retrosynthetic strategies, as well as the execution of previously unattainable tactical maneuvers. Finally, shortcomings of existing processes are highlighted; this Review illustrates how some highlighted total syntheses can be further economized by adopting next-generation ligand-enabled approaches.
Keywords: C−H activation; C−H functionalization; catalysis; total synthesis; transition metals.
© 2020 Wiley-VCH GmbH.
Figures

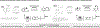
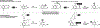




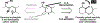
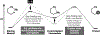
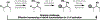
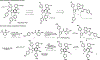

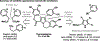

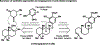
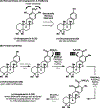



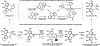
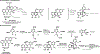

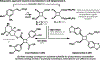

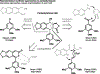

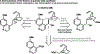
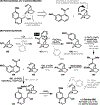

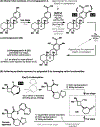


References
-
- Wöhler F, Ann. Phys 1828, 88, 253–256.
-
- Robinson R, J. Chem. Soc. Trans 1917, 111, 762–768.
-
- Sheehan JC, Henbry-Logan KR, J. Am. Chem. Soc 1957, 79, 1262–1263.
-
- Eschenmoser A, Wintner CE, Science 1977, 196, 1410–1420. - PubMed
-
- Nicolaou KC, Petasis NA, Zipkin RE, Uenishi J, J. Am. Chem. Soc 1982, 104, 5555–5557.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

