Amino acid synthesis loss in parasitoid wasps and other hymenopterans
- PMID: 33074103
- PMCID: PMC7593089
- DOI: 10.7554/eLife.59795
Amino acid synthesis loss in parasitoid wasps and other hymenopterans
Retraction in
-
Retraction: Amino acid synthesis loss in parasitoid wasps and other hymenopterans.Elife. 2020 Dec 1;9:e65123. doi: 10.7554/eLife.65123. Elife. 2020. PMID: 33258773 Free PMC article.
Abstract
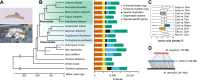
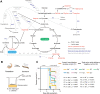
Insects utilize diverse food resources which can affect the evolution of their genomic repertoire, including leading to gene losses in different nutrient pathways. Here, we investigate gene loss in amino acid synthesis pathways, with special attention to hymenopterans and parasitoid wasps. Using comparative genomics, we find that synthesis capability for tryptophan, phenylalanine, tyrosine, and histidine was lost in holometabolous insects prior to hymenopteran divergence, while valine, leucine, and isoleucine were lost in the common ancestor of Hymenoptera. Subsequently, multiple loss events of lysine synthesis occurred independently in the Parasitoida and Aculeata. Experiments in the parasitoid Cotesia chilonis confirm that it has lost the ability to synthesize eight amino acids. Our findings provide insights into amino acid synthesis evolution, and specifically can be used to inform the design of parasitoid artificial diets for pest control.
Keywords: Cotesia chilonis; amino acid synthesis; comparative genomics; genetics; genome sequencing; genomics; parasitoid wasps; trait loss.
© 2020, Ye et al.
Conflict of interest statement
XY, SX, ZT, YY, JW, KY, HW, YM, ZY, SC, CY, FW, HY, QF, QS, JW, GY, FL No competing interests declared
Figures








Similar articles
-
Genome of the parasitoid wasp Cotesia chilonis sheds light on amino acid resource exploitation.BMC Biol. 2022 May 24;20(1):118. doi: 10.1186/s12915-022-01313-3. BMC Biol. 2022. PMID: 35606775 Free PMC article.
-
A Chromosome-Level Genome Assembly of the Parasitoid Wasp, Cotesia glomerata (Hymenoptera: Braconidae).J Hered. 2021 Nov 1;112(6):558-564. doi: 10.1093/jhered/esab032. J Hered. 2021. PMID: 34043785
-
Screening Sugars Can Benefit the Parasitoid Cotesia chilonis (Hymenoptera: Braconidae) Without Benefiting Its Host, Chilo suppressalis (Lepidoptera: Crambidae).J Econ Entomol. 2019 Sep 23;112(5):2142-2148. doi: 10.1093/jee/toz166. J Econ Entomol. 2019. PMID: 31219581
-
Dietary Challenges for Parasitoid Wasps (Hymenoptera: Ichneumonoidea); Coping with Toxic Hosts, or Not?Toxins (Basel). 2023 Jun 29;15(7):424. doi: 10.3390/toxins15070424. Toxins (Basel). 2023. PMID: 37505693 Free PMC article. Review.
-
Biodiversity of hymenopteran parasitoids.Curr Opin Insect Sci. 2023 Apr;56:101026. doi: 10.1016/j.cois.2023.101026. Epub 2023 Mar 24. Curr Opin Insect Sci. 2023. PMID: 36966863 Review.
References
-
- Bank S, Sann M, Mayer C, Meusemann K, Donath A, Podsiadlowski L, Kozlov A, Petersen M, Krogmann L, Meier R, Rosa P, Schmitt T, Wurdack M, Liu S, Zhou X, Misof B, Peters RS, Niehuis O. Transcriptome and target DNA enrichment sequence data provide new insights into the phylogeny of vespid wasps (Hymenoptera: aculeata: vespidae) Molecular Phylogenetics and Evolution. 2017;116:213–226. doi: 10.1016/j.ympev.2017.08.020. - DOI - PubMed
Publication types
Grants and funding
- Major International (Regional) Joint Research Project of NSFC,31620103915/National Natural Science Foundation of China/International
- Key Program of National Natural Science Foundation of China,31830074/National Natural Science Foundation of China/International
- 31772238/National Natural Science Foundation of China/International
- 2017YFD0200400/National Key Research and Development Program of China/International
- 2017YFD0200904/National Key Research and Development Program of China/International
- IOS-1456233/National Science Foundation/International
- Major International (Regional) Joint Research Project of National Natural Science Foundation of China 31620103915/National Natural Science Foundation of China/International
- Key Program of National Natural Science Foundation of China 31830074/National Natural Science Foundation of China/International
LinkOut - more resources
Full Text Sources

