Targeting interleukin-17 receptor B enhances gemcitabine sensitivity through downregulation of mucins in pancreatic cancer
- PMID: 33082357
- PMCID: PMC7576602
- DOI: 10.1038/s41598-020-73659-z
Targeting interleukin-17 receptor B enhances gemcitabine sensitivity through downregulation of mucins in pancreatic cancer
Abstract
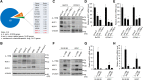
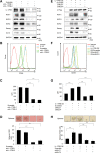
Pancreatic cancer is the fourth leading cause of death worldwide due to its poorest prognoses with a 7% 5-year survival rate. Eighty percent of pancreatic cancer patients relapse after chemotherapy and develop early metastasis and drug resistance. Resistance to nucleoside analog gemcitabine frequently used in first-line therapy is an urgent issue in pancreatic cancer treatment. Expression of mucin (MUC) glycoproteins has been shown to enhance chemoresistance via increased cell stemness. Here we show interlukine-17 receptor B (IL-17RB) expression is positively correlated with MUC1 and MUC4 expression in pancreatic cancer cells and tumor tissue. Moreover, IL-17RB transcriptionally up-regulates expression of MUC1 and MUC4 to enhance cancer stem-like properties and resistance to gemcitabine. These results suggest IL-17RB can be a potential target for pancreatic cancer therapy. Indeed, treatment with IL-17RB-neutralizing antibody has a synergistic effect in combination with gemcitabine for killing pancreatic cancer cells. Altogether, these findings provide feasible applications for IL-17RB-targeting therapy in pancreatic cancer treatment.
Conflict of interest statement
The authors declare no competing interests.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

