The Janus-like role of proline metabolism in cancer
- PMID: 33083024
- PMCID: PMC7560826
- DOI: 10.1038/s41420-020-00341-8
The Janus-like role of proline metabolism in cancer
Abstract
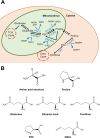
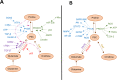
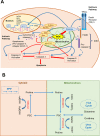
The metabolism of the non-essential amino acid L-proline is emerging as a key pathway in the metabolic rewiring that sustains cancer cells proliferation, survival and metastatic spread. Pyrroline-5-carboxylate reductase (PYCR) and proline dehydrogenase (PRODH) enzymes, which catalyze the last step in proline biosynthesis and the first step of its catabolism, respectively, have been extensively associated with the progression of several malignancies, and have been exposed as potential targets for anticancer drug development. As investigations into the links between proline metabolism and cancer accumulate, the complexity, and sometimes contradictory nature of this interaction emerge. It is clear that the role of proline metabolism enzymes in cancer depends on tumor type, with different cancers and cancer-related phenotypes displaying different dependencies on these enzymes. Unexpectedly, the outcome of rewiring proline metabolism also differs between conditions of nutrient and oxygen limitation. Here, we provide a comprehensive review of proline metabolism in cancer; we collate the experimental evidence that links proline metabolism with the different aspects of cancer progression and critically discuss the potential mechanisms involved.
Keywords: Cancer; Cancer metabolism.
© The Author(s) 2020.
Conflict of interest statement
Conflict of interestThe authors declare that they have no conflict of interest.
Figures
Similar articles
-
Oncogenic human herpesvirus hijacks proline metabolism for tumorigenesis.Proc Natl Acad Sci U S A. 2020 Apr 7;117(14):8083-8093. doi: 10.1073/pnas.1918607117. Epub 2020 Mar 25. Proc Natl Acad Sci U S A. 2020. PMID: 32213586 Free PMC article.
-
Proline metabolism in cancer.Amino Acids. 2021 Dec;53(12):1769-1777. doi: 10.1007/s00726-021-03060-1. Epub 2021 Aug 14. Amino Acids. 2021. PMID: 34390414 Review.
-
Increased mitochondrial proline metabolism sustains proliferation and survival of colorectal cancer cells.PLoS One. 2022 Feb 7;17(2):e0262364. doi: 10.1371/journal.pone.0262364. eCollection 2022. PLoS One. 2022. PMID: 35130302 Free PMC article.
-
Structure, biochemistry, and gene expression patterns of the proline biosynthetic enzyme pyrroline-5-carboxylate reductase (PYCR), an emerging cancer therapy target.Amino Acids. 2021 Dec;53(12):1817-1834. doi: 10.1007/s00726-021-02999-5. Epub 2021 May 18. Amino Acids. 2021. PMID: 34003320 Free PMC article. Review.
-
The Proline Cycle As a Potential Cancer Therapy Target.Biochemistry. 2018 Jun 26;57(25):3433-3444. doi: 10.1021/acs.biochem.8b00215. Epub 2018 Apr 23. Biochemistry. 2018. PMID: 29648801 Free PMC article.
Cited by
-
Hallmarks of Metabolic Reprogramming and Their Role in Viral Pathogenesis.Viruses. 2022 Mar 14;14(3):602. doi: 10.3390/v14030602. Viruses. 2022. PMID: 35337009 Free PMC article. Review.
-
Schizophrenia-Like Behaviors Arising from Dysregulated Proline Metabolism Are Associated with Altered Neuronal Morphology and Function in Mice with Hippocampal PRODH Deficiency.Aging Dis. 2024 Aug 1;15(4):1952-1968. doi: 10.14336/AD.2023.0902. Aging Dis. 2024. PMID: 37815900 Free PMC article.
-
The Role of Non-essential Amino Acids in T Cell Function and Anti-tumour Immunity.Arch Immunol Ther Exp (Warsz). 2021 Oct 12;69(1):29. doi: 10.1007/s00005-021-00633-6. Arch Immunol Ther Exp (Warsz). 2021. PMID: 34637000 Free PMC article. Review.
-
Perspectives, past, present and future: the proline cycle/proline-collagen regulatory axis.Amino Acids. 2021 Dec;53(12):1967-1975. doi: 10.1007/s00726-021-03103-7. Epub 2021 Nov 26. Amino Acids. 2021. PMID: 34825974 Free PMC article.
-
P5C as an Interface of Proline Interconvertible Amino Acids and Its Role in Regulation of Cell Survival and Apoptosis.Int J Mol Sci. 2021 Oct 29;22(21):11763. doi: 10.3390/ijms222111763. Int J Mol Sci. 2021. PMID: 34769188 Free PMC article. Review.

