Hydrogen sulfide negatively regulates cd-induced cell death in cucumber (Cucumis sativus L) root tip cells
- PMID: 33087071
- PMCID: PMC7579943
- DOI: 10.1186/s12870-020-02687-8
Hydrogen sulfide negatively regulates cd-induced cell death in cucumber (Cucumis sativus L) root tip cells
Abstract
Background: Hydrogen sulfide (H2S) is a gas signal molecule involved in regulating plants tolerance to heavy metals stress. In this study, we investigated the role of H2S in cadmium-(Cd-) induced cell death of root tips of cucumber seedlings.
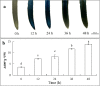
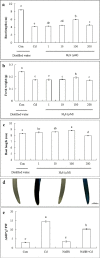
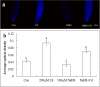
Results: The results showed that the application of 200 μM Cd caused cell death, increased the content of reactive oxygen species (ROS), chromatin condensation, the release of Cytochrome c (Cyt c) from mitochondria and activated caspase-3-like protease. Pretreatment of seedlings with 100 μM sodium hydrogen sulfide (NaHS, a H2S donor) effectively alleviated the growth inhibition and reduced cell death of root tips caused by Cd stress. Additionally, NaHS + Cd treatment could decrease the ROS level and enhanced antioxidant enzyme activity. Pretreatment with NaHS also inhibited the release of Cyt c from the mitochondria, the opening of the mitochondrial permeability transition pore (MPTP), and the activity of caspase-3-like protease in the root tips of cucumber seedling under Cd stress.
Conclusion: H2S inhibited Cd-induced cell death in cucumber root tips by reducing ROS accumulation, activating the antioxidant system, inhibiting mitochondrial Cyt c release and reducing the opening of the MPTP. The results suggest that H2S is a negative regulator of Cd-induced cell death in the root tips of cucumber seedling.
Keywords: Caspase-3-like protease; Cell death; Cyt c; Mitochondria; Oxidative damage.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures





Similar articles
-
Methyl Jasmonate Was Involved in Hydrogen Sulfide-Alleviated Cadmium Stress in Cucumber Plants Through ROS Homeostasis and Chlorophyll Metabolism.Int J Mol Sci. 2025 Jan 8;26(2):475. doi: 10.3390/ijms26020475. Int J Mol Sci. 2025. PMID: 39859190 Free PMC article.
-
Hydrogen sulfide alleviates oxidative damage under excess nitrate stress through MAPK/NO signaling in cucumber.Plant Physiol Biochem. 2019 Feb;135:1-8. doi: 10.1016/j.plaphy.2018.11.017. Epub 2018 Nov 15. Plant Physiol Biochem. 2019. PMID: 30481610
-
Interaction between the signaling molecules hydrogen sulfide and hydrogen peroxide and their role in vacuolar H+ -ATPase regulation in cadmium-stressed cucumber roots.Physiol Plant. 2019 Jun;166(2):688-704. doi: 10.1111/ppl.12819. Epub 2018 Sep 28. Physiol Plant. 2019. PMID: 30120777
-
Boron toxicity is alleviated by hydrogen sulfide in cucumber (Cucumis sativus L.) seedlings.Planta. 2010 May;231(6):1301-9. doi: 10.1007/s00425-010-1134-9. Epub 2010 Mar 12. Planta. 2010. PMID: 20224946
-
Auxin acts as a downstream signaling molecule involved in hydrogen sulfide-induced chilling tolerance in cucumber.Planta. 2020 Feb 19;251(3):69. doi: 10.1007/s00425-020-03362-w. Planta. 2020. PMID: 32076872
Cited by
-
2,4-Epibrassinolide Mitigates Cd Stress by Enhancing Chloroplast Structural Remodeling and Chlorophyll Metabolism in Vigna angularis Leaves.Biology (Basel). 2025 Jun 10;14(6):674. doi: 10.3390/biology14060674. Biology (Basel). 2025. PMID: 40563925 Free PMC article.
-
Foliar Spraying of NaHS Alleviates Cucumber Salt Stress by Maintaining N+/K+ Balance and Activating Salt Tolerance Signaling Pathways.Plants (Basel). 2023 Jun 26;12(13):2450. doi: 10.3390/plants12132450. Plants (Basel). 2023. PMID: 37447010 Free PMC article.
-
RNA-Seq Identification of Cd Responsive Transporters Provides Insights into the Association of Oxidation Resistance and Cd Accumulation in Cucumis sativus L.Antioxidants (Basel). 2021 Dec 10;10(12):1973. doi: 10.3390/antiox10121973. Antioxidants (Basel). 2021. PMID: 34943077 Free PMC article.
-
Syntaxin of plants71 plays essential roles in plant development and stress response via regulating pH homeostasis.Front Plant Sci. 2023 Jun 5;14:1198353. doi: 10.3389/fpls.2023.1198353. eCollection 2023. Front Plant Sci. 2023. PMID: 37342145 Free PMC article.
-
Gm6PGDH1, a Cytosolic 6-Phosphogluconate Dehydrogenase, Enhanced Tolerance to Phosphate Starvation by Improving Root System Development and Modifying the Antioxidant System in Soybean.Front Plant Sci. 2021 Aug 13;12:704983. doi: 10.3389/fpls.2021.704983. eCollection 2021. Front Plant Sci. 2021. PMID: 34484268 Free PMC article.
References
-
- Abedi E: Cadmium stress in rice plants: The effect of cadmium on seed germination and seedling growth of rice plant (Oriza sativa L.). 2019.
-
- Gallego SM, Pena LB, Barcia RA, Azpilicueta CE, Iannone MF, Rosales EP, Zawoznik MS, Groppa MD, Benavides MP. Unravelling cadmium toxicity and tolerance in plants: insight into regulatory mechanisms. Environ Exp Bot. 2012;83(5):33–46. doi: 10.1016/j.envexpbot.2012.04.006. - DOI
MeSH terms
Substances
Grants and funding
- 2018YFD0201205/the National key research and development projects
- No. 31660584/the National Natural Science Foundation of China
- CARS-23-C-07/China Agriculture Research System
- No.17ZD2NA015/Gansu Province Science and Technology Key Project Fund
- 1610RJZA098/Natural Science Foundation of Gansu References Province, China
LinkOut - more resources
Full Text Sources
Research Materials

