A Straightforward Synthesis of Polyketides via Ester Dienolate Matteson Homologation
- PMID: 33089903
- PMCID: PMC7839490
- DOI: 10.1002/chem.202004650
A Straightforward Synthesis of Polyketides via Ester Dienolate Matteson Homologation
Abstract
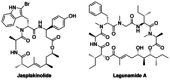
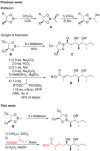
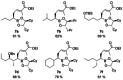
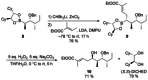
Application of ester dienolates as nucleophiles in Matteson homologations allows for the stereoselective synthesis of highly substituted α,β-unsaturated δ-hydroxy carboxyl acids, structural motifs widespread found in polyketide natural products. The protocol is rather flexible and permits the introduction of substituents and functionalities also at those positions which are not accessible by the commonly used aldol reaction. Therefore, this ester dienolate Matteson approach is an interesting alternative to the "classical" polyketide syntheses.
Keywords: Matteson homologation; boronic esters; enolates; natural products; polyketides.
© 2020 The Authors. Published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Syntheses of Marine Natural Products via Matteson Homologations and Related Processes.Mar Drugs. 2025 Jan 2;23(1):20. doi: 10.3390/md23010020. Mar Drugs. 2025. PMID: 39852522 Free PMC article. Review.
-
Stereoselective Synthesis of Secondary and Tertiary Boronic Esters via Matteson Homologation.Org Lett. 2023 Sep 22;25(37):6835-6839. doi: 10.1021/acs.orglett.3c02360. Epub 2023 Sep 11. Org Lett. 2023. PMID: 37695257
-
Application of Vinyl Nucleophiles in Matteson Homologations.Org Lett. 2022 May 27;24(20):3599-3603. doi: 10.1021/acs.orglett.2c01119. Epub 2022 May 18. Org Lett. 2022. PMID: 35583355
-
Stereoselective Syntheses of Highly Substituted Tetrahydrofurans based on Matteson Homologations.Chemistry. 2025 Apr 15;31(22):e202500560. doi: 10.1002/chem.202500560. Epub 2025 Mar 12. Chemistry. 2025. PMID: 40072278
-
Modern aldol methods for the total synthesis of polyketides.Angew Chem Int Ed Engl. 2006 Nov 20;45(45):7506-25. doi: 10.1002/anie.200602780. Angew Chem Int Ed Engl. 2006. PMID: 17103481 Review.
Cited by
-
Synthesis and Late-Stage Modification of (-)-Doliculide Derivatives Using Matteson's Homologation Approach.Mar Drugs. 2024 Apr 8;22(4):165. doi: 10.3390/md22040165. Mar Drugs. 2024. PMID: 38667782 Free PMC article.
-
Syntheses of Marine Natural Products via Matteson Homologations and Related Processes.Mar Drugs. 2025 Jan 2;23(1):20. doi: 10.3390/md23010020. Mar Drugs. 2025. PMID: 39852522 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources