INEXAS: A Phase 2 Randomized Trial of On-demand Inhaled Interferon Beta-1a in Severe Asthmatics
- PMID: 33091192
- PMCID: PMC7984268
- DOI: 10.1111/cea.13765
INEXAS: A Phase 2 Randomized Trial of On-demand Inhaled Interferon Beta-1a in Severe Asthmatics
Abstract
Background: Upper respiratory tract infections (URTIs) are important triggers for asthma exacerbations. We hypothesized that inhalation of the anti-viral cytokine, interferon (IFN)-β, during URTI, could prevent these exacerbations.
Objective: To evaluate the efficacy of on-demand inhaled IFN-β1a (AZD9412) to prevent severe asthma exacerbations following symptomatic URTI.
Methods: This was a randomized, double-blind, placebo-controlled trial in which patients with severe asthma (GINA 4-5; n = 121) reporting URTI symptoms were randomized to 14 days of once-daily nebulized AZD9412 or placebo. The primary endpoint was severe exacerbations during treatment. Secondary endpoints included 6-item asthma control questionnaire (ACQ-6) and lung function. Exploratory biomarkers included IFN-response markers in serum and sputum, blood leucocyte counts and serum inflammatory cytokines.
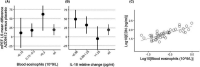
Results: Following a pre-planned interim analysis, the trial was terminated early due to an unexpectedly low exacerbation rate. Asthma worsenings were generally mild and tended to peak at randomization, possibly contributing to the lack of benefit of AZD9412 on other asthma endpoints. Numerically, AZD9412 did not reduce severe exacerbation rate, ACQ-6, asthma symptom scores or reliever medication use. AZD9412 improved lung function (morning peak expiratory flow; mPEF) by 19.7 L/min. Exploratory post hoc analyses indicated a greater mPEF improvement by AZD9412 in patients with high blood eosinophils (>0.3 × 109 /L) at screening and low serum interleukin-18 relative change at pre-treatment baseline. Pharmacodynamic effect of AZD9412 was confirmed using IFN-response markers.
Conclusions & clinical relevance: Colds did not have the impact on asthma patients that was expected and, due to the low exacerbation rate, the trial was stopped early. On-demand AZD9412 treatment did not numerically reduce the number of exacerbations, but did attenuate URTI-induced worsening of mPEF. Severe asthma patients with high blood eosinophils or low serum interleukin-18 response are potential subgroups for further investigation of inhaled IFN-β1a.
Trial registration: ClinicalTrials.gov NCT02491684.
Keywords: IFN response; IL-18; asthma; eosinophils; exacerbation; interferon; viral URTI.
@2020 AstraZeneca. International Journal of Cosmetic Science published by John Wiley & Sons.
Conflict of interest statement
CM, MO, PG, AM, MA, MF, CAD, AC, JP, KK and CW are employees and shareholders of AstraZeneca. MK is a former employee of AstraZeneca. PM and RM are directors and shareholders in Synairgen plc. TH reports personal fees and non‐financial support from AstraZeneca and GlaxoSmithKline, and personal fees from Chiesi and Vectura, outside of the submitted work.
Figures




Similar articles
-
The effect of inhaled IFN-β on worsening of asthma symptoms caused by viral infections. A randomized trial.Am J Respir Crit Care Med. 2014 Jul 15;190(2):145-54. doi: 10.1164/rccm.201312-2235OC. Am J Respir Crit Care Med. 2014. PMID: 24937476 Free PMC article. Clinical Trial.
-
[Standard technical specifications for methacholine chloride (Methacholine) bronchial challenge test (2023)].Zhonghua Jie He He Hu Xi Za Zhi. 2024 Feb 12;47(2):101-119. doi: 10.3760/cma.j.cn112147-20231019-00247. Zhonghua Jie He He Hu Xi Za Zhi. 2024. PMID: 38309959 Chinese.
-
Efficacy and safety of tralokinumab in patients with severe uncontrolled asthma: a randomised, double-blind, placebo-controlled, phase 2b trial.Lancet Respir Med. 2015 Sep;3(9):692-701. doi: 10.1016/S2213-2600(15)00197-6. Epub 2015 Jul 28. Lancet Respir Med. 2015. PMID: 26231288 Clinical Trial.
-
Tiotropium bromide. A review of its use as maintenance therapy in patients with COPD.Treat Respir Med. 2004;3(4):247-68. doi: 10.2165/00151829-200403040-00005. Treat Respir Med. 2004. PMID: 15350163 Review.
-
Impact of baseline clinical asthma characteristics on the response to mepolizumab: a post hoc meta-analysis of two Phase III trials.Respir Res. 2021 Jun 22;22(1):184. doi: 10.1186/s12931-021-01767-z. Respir Res. 2021. PMID: 34158028 Free PMC article. Review.
Cited by
-
Airway Epithelial Innate Immunity.Front Physiol. 2021 Nov 26;12:749077. doi: 10.3389/fphys.2021.749077. eCollection 2021. Front Physiol. 2021. PMID: 34899381 Free PMC article.
-
Deciphering Respiratory-Virus-Associated Interferon Signaling in COPD Airway Epithelium.Medicina (Kaunas). 2022 Jan 13;58(1):121. doi: 10.3390/medicina58010121. Medicina (Kaunas). 2022. PMID: 35056429 Free PMC article. Review.
-
Nebulised interferon beta-1a (SNG001) in the treatment of viral exacerbations of COPD.Respir Res. 2024 May 29;25(1):228. doi: 10.1186/s12931-024-02854-7. Respir Res. 2024. PMID: 38811970 Free PMC article. Clinical Trial.
-
New targets for type 2-low asthma.Korean J Intern Med. 2024 Mar;39(2):215-227. doi: 10.3904/kjim.2023.299. Epub 2024 Feb 6. Korean J Intern Med. 2024. PMID: 38317271 Free PMC article. Review.
-
Can Allergen Immunotherapy Improve Antiviral Immunity in Patients with Allergic Asthma?Am J Respir Crit Care Med. 2023 May 1;207(9):1112-1114. doi: 10.1164/rccm.202302-0234ED. Am J Respir Crit Care Med. 2023. PMID: 36812499 Free PMC article. No abstract available.

