Early Changes in Kidney Transplant Immunosuppression Regimens During the COVID-19 Pandemic
- PMID: 33093404
- PMCID: PMC7752821
- DOI: 10.1097/TP.0000000000003502
Early Changes in Kidney Transplant Immunosuppression Regimens During the COVID-19 Pandemic
Abstract
Background: Kidney transplant recipients have higher risk of infectious diseases due to their reliance on immunosuppression. During the current COVID-19 pandemic, some clinicians might have opted for less potent immunosuppressive agents to counterbalance the novel infectious risk. We conducted a nationwide study to characterize immunosuppression use and subsequent clinical outcomes during the first 5 months of COVID-19 pandemic in the United States.
Methods: Using data from the Scientific Registry of Transplant Recipients, we studied all kidney-only recipients in the United States from January 1, 2017, to March 12, 2020 ("prepandemic" era; n = 64 849) and from March 13, 2020, to July 31, 2020 ("pandemic" era; n = 5035). We compared the use of lymphocyte-depleting agents (versus basiliximab or no induction) and maintenance steroids (versus steroid avoidance/withdrawal) in the pandemic era compared with the prepandemic era. Then, we compared early posttransplant outcomes by immunosuppression regimen during the pandemic era.
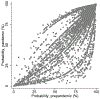
Results: Recipients in the pandemic era were substantially less likely to receive lymphocyte-depleting induction agents compared with their prepandemic counterparts (aOR = 0.400.530.69); similar trends were found across subgroups of state-level COVID-19 incidence, donor type, and recipient age. However, lymphocyte-depleting induction agents were associated with decreased rejection during admission (aOR = 0.110.230.47) but not with increased mortality in the pandemic era (aHR = 0.130.471.66). On the other hand, the use of maintenance steroids versus early steroid withdrawal remained similar (aOR = 0.711.071.62).
Conclusions: The use of lymphocyte-depleting induction agents has decreased in favor of basiliximab and no induction during the COVID-19 pandemic. However, this shift might have resulted in increases in rejection with no clear reductions in posttransplant mortality.
Copyright © 2020 Wolters Kluwer Health, Inc. All rights reserved.
Conflict of interest statement
D.L.S. receives speaking honoraria from Sanofi, Novartis, and CSL Behring. All other authors declare no conflicts of interest.
Figures

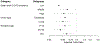
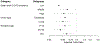
Comment in
-
A case report of successful kidney transplantation from a deceased donor with terminal COVID-19-related lung damage: Ongoing dilemma between discarding and accepting organs in COVID-19 era!Transpl Infect Dis. 2021 Oct;23(5):e13683. doi: 10.1111/tid.13683. Epub 2021 Jul 12. Transpl Infect Dis. 2021. PMID: 34227197 Free PMC article. No abstract available.
References
-
- Brennan DC, Daller JA, Lake KD, et al. Rabbit Antithymocyte Globulin versus Basiliximab in Renal Transplantation. N Engl J Med. 2006;355(19):1967–1977. - PubMed
-
- Woodle ES, First MR, Pirsch J, et al. A prospective, randomized, double-blind, placebo-controlled multicenter trial comparing early (7 day) corticosteroid cessation versus long-term, low-dose corticosteroid therapy. Ann Surg. 2008;248(4):564–577. - PubMed
-
- Fishman JA. Infection in Solid-Organ Transplant Recipients. N Engl J Med. 2007;357(25):2601–2614. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

