Intravenous immunoglobulins as first-line treatment in idiopathic inflammatory myopathies: a pilot study
- PMID: 33099648
- PMCID: PMC8023983
- DOI: 10.1093/rheumatology/keaa459
Intravenous immunoglobulins as first-line treatment in idiopathic inflammatory myopathies: a pilot study
Abstract
Objectives: We explored efficacy and safety of IVIg as first-line treatment in patients with an idiopathic inflammatory myopathy.
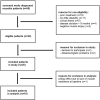
Methods: In this investigator-initiated phase 2 open-label study, we included 20 adults with a newly diagnosed, biopsy-proven idiopathic inflammatory myopathy, and a disease duration of less than 9 months. Patients with IBM and prior use of immunosuppressants were excluded. The standard treatment regimen consisted of IVIg (Privigen) monotherapy for 9 weeks: a loading dose (2 g/kg body weight) and two subsequent maintenance doses (1 g/kg body weight) with a 3-week interval. The primary outcome was the number of patients with at least moderate improvement on the 2016 ACR/EULAR Total Improvement Score. Secondary outcomes included time to improvement, the number of patients requiring rescue medication and serious adverse events.
Results: We included patients with DM (n = 9), immune-mediated necrotizing myopathy (n = 6), non-specific myositis/overlap myositis (n = 4) and anti-synthetase syndrome (n = 1). One patient was excluded from analyses because of minimal weakness resulting in a ceiling effect. Eight patients (8/19 = 42.0%; Clopper-Pearson 95% CI: 19.6, 64.6) had at least moderate improvement by 9 weeks. Of these, six reached improvement by 3 weeks. Seven patients required rescue medication due to insufficient efficacy and prematurely ended the study. Three serious adverse events occurred, of which one was pulmonary embolism.
Conclusion: First-line IVIg monotherapy led to at least moderate improvement in nearly half of patients with a fast clinical response in the majority of responders.
Trial registration: Netherlands Trial Register identifier, NTR6160.
Keywords: immunotherapy; myositis and muscle disease.
© The Author(s) 2020. Published by Oxford University Press on behalf of the British Society for Rheumatology.
Figures


References
-
- Van de Vlekkert J, Hoogendijk JE, de Visser M.. Long-term follow-up of 62 patients with myositis. J Neurol 2014;261:992–8. - PubMed
-
- Marie I. Morbidity and mortality in adult polymyositis and dermatomyositis. Curr Rheumatol Rep 2012;14:275–85. - PubMed
-
- Van de Vlekkert J, Hoogendijk JE, de Haan RJ. et al. Oral dexamethasone pulse therapy versus daily prednisolone in sub-acute onset myositis, a randomised clinical trial. Neuromuscul Disord 2010;20:382–9. - PubMed
-
- Dalakas MC. Intravenous immunoglobulin in autoimmune neuromuscular diseases. JAMA 2004;291:2367–75. - PubMed
-
- Dalakas MC, Illa I, Dambrosia JM. et al. A controlled trial of high-dose intravenous immune globulin infusions as treatment for dermatomyositis. N Engl J Med 1993;329:1993–2000. - PubMed

