Splenectomy Modulates the Erythrocyte Turnover and Basigin (CD147) Expression in Mice
- PMID: 33100715
- PMCID: PMC7573055
- DOI: 10.1007/s12288-020-01272-1
Splenectomy Modulates the Erythrocyte Turnover and Basigin (CD147) Expression in Mice
Abstract
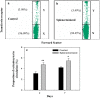
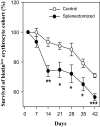
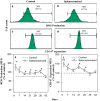
The present study was designed to study the splenectomy induced modulation of erythrocyte turnover in mice. We have also studied the modulation of reactive oxygen species (ROS) and basigin (CD147) expression level on erythrocytes in splenectomized condition. The erythrocyte turnover was studied by a newly developed double in vivo biotinylation (DIB) technique. This technique enables to discriminate three different age (young, intermediate and old) groups of erythrocytes. The expression level of ROS and CD147 was studied by staining with CM-H2DCFDA stain and anti-mouse CD147 monocloclonal antibody followed by flow cytometry. We observed that intermediate and old age groups of erythrocytes were randomly eliminated in splenectomized condition. A marked surge in the blood reticulocyte count was observed in splenectomized mice. Splenectomy induced the level of ROS and CD147 expression on erythrocytes. The expression level of ROS was induced up to 35 days, but it reversed to basal level by 42 days indicating the emergence of refractoriness to splenectomy. The CD147 expression was significantly higher on day 7, 21 and 28 but it also normalizes on later time points. We conclude that erythrocyte turnover is significantly modulated in splenectomized mice. The enhanced level of ROS and CD147 expression may be a possible cause to increase erythrocyte removal in splenectomized mice.
Keywords: Basigin; Double in vivo biotinylation; Erythrocyte; Flow cytometry; Reactive oxygen species; Splenectomy.
© Indian Society of Hematology and Blood Transfusion 2020.
Conflict of interest statement
Conflict of interestThe authors declare no conflict of interest.
Figures

References
LinkOut - more resources
Full Text Sources
