Rhein Protects Against Neurological Deficits After Traumatic Brain Injury in Mice via Inhibiting Neuronal Pyroptosis
- PMID: 33101024
- PMCID: PMC7554525
- DOI: 10.3389/fphar.2020.564367
Rhein Protects Against Neurological Deficits After Traumatic Brain Injury in Mice via Inhibiting Neuronal Pyroptosis
Abstract
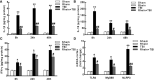
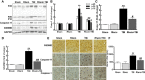
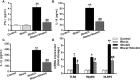
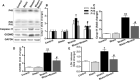
Neurological dysfunction provoked by traumatic brain injury (TBI) makes a huge impact on individual learning ability, memory level, social participation, and quality of life. Pyroptosis, the caspase-1-dependent cell death, which is associated with the release of numerous pro-inflammatory factors, plays a major role in the pathological process after TBI. Inhibition of pyroptosis has been shown to be an attractive strategy for the treatment of various neurological disorders. Here, we found that Rhein, an anthraquinone derived from the medicinal plant rhubarb, attenuated TBI-induced upregulation of pro-inflammatory cytokines, blood lactate dehydrogenase (LDH), and pyroptosis-related proteins, as well as reduced neurological dysfunction in a mouse TBI model. Consistently, Rhein inhibitd equiaxial stretch-induced neuron pyroptosis, LDH release, and upregulation of pro-inflammatory factors in vitro. Thus, our study suggested that Rhein protected against neurological deficits after TBI via inhibiting neuronal pyroptosis.
Keywords: Rhein; inflammatory cytokines; neurological deficits; pyroptosis; traumatic brain injury.
Copyright © 2020 Bi, Ma, Ji, Chang, Liu and Xie.
Figures

References
LinkOut - more resources
Full Text Sources

