Autophagy and Inflammation Regulation in Acute Kidney Injury
- PMID: 33101057
- PMCID: PMC7546328
- DOI: 10.3389/fphys.2020.576463
Autophagy and Inflammation Regulation in Acute Kidney Injury
Erratum in
-
Corrigendum: Autophagy and Inflammation Regulation in Acute Kidney Injury.Front Physiol. 2021 Feb 9;11:639938. doi: 10.3389/fphys.2020.639938. eCollection 2020. Front Physiol. 2021. PMID: 33633580 Free PMC article.
Abstract
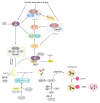
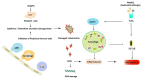
Autophagy at an appropriate juncture in the cell cycle exerts protective effects in acute kidney injury (AKI), whereas abnormal autophagy may lead to cell death. Inflammatory response plays a pivotal role in the pathophysiological process of kidney injury and repair during AKI. Several studies have reported an interaction between autophagy and inflammation in the pathogenesis of AKI. This review outlines recent advances in the investigation of the role of autophagy in inflammatory response regulation based on the following aspects. (1) Autophagy inhibits inflammatory responses induced in AKI through the regulation of mTOR and AMPK pathways and the inhibition of inflammasomes activation. (2) Autophagy can also help in the regulation of inflammatory responses through the nuclear factor kappa B pathway, which is beneficial to the recovery of kidney tissues. These studies reviewed here provide better insight into the mechanisms underlying the protective effects of the autophagy-inflammatory pathway. Through this review, we suggest that the autophagy-inflammatory pathway may serve as an alternative target for the treatment of AKI.
Keywords: acute kidney injury; autophagy; immune cells; inflammatory; tubular epithelial cells.
Copyright © 2020 Gong, Pan and Yang.
Figures

References
-
- Abdallah N. H., Baulies A., Bouhlel A., Bejaoui M., Zaouali M. A., Mimouna S. B., et al. . (2018). Zinc mitigates renal ischemia-reperfusion injury in rats by modulating oxidative stress, endoplasmic reticulum stress, and autophagy. J. Cell. Physiol. 233, 8677–8690. 10.1002/jcp.26747, PMID: - DOI - PubMed
-
- Bao D. S., Wu Y. K., Fu S. J., Wang G. Y., Yang S. J., Liang G. B., et al. . (2017). Hyperbaric oxygenation protects against ischemia- reperfusion injury in transplanted rat kidneys by triggering autophagy and inhibiting inflammatory response. Ann. Transplant. 22, 75–82. 10.12659/aot.901102, PMID: - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

