Unstimulated Serum Thyroglobulin Levels after Thyroidectomy and Radioiodine Therapy for Intermediate-Risk Thyroid Cancer Are Not Always a Reliable Marker of Lymph Node Recurrence: Case Report and a Lesson for Clinicians
- PMID: 33101736
- PMCID: PMC7568143
- DOI: 10.1155/2020/8827503
Unstimulated Serum Thyroglobulin Levels after Thyroidectomy and Radioiodine Therapy for Intermediate-Risk Thyroid Cancer Are Not Always a Reliable Marker of Lymph Node Recurrence: Case Report and a Lesson for Clinicians
Abstract
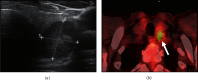
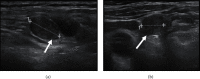
Over 50% of patients with papillary thyroid carcinoma (PTC) have cervical lymph-node metastasis on diagnosis, and up to 30% show nodal recurrence after surgery plus radioactive iodine (131I) (RAI) therapy. The combination of ultrasonography (US) and fine-needle aspiration cytology (FNAC) and the measurement of thyroglobulin (Tg) in washout fluid are cornerstones in the diagnosis of nodal metastasis. In the absence of anti-Tg antibodies, unstimulated serum thyroglobulin (Tg) levels are generally a reliable marker of recurrent disease, and 18F-FDG positron emission tomography (PET)/computed tomography (CT) plays an important role in the imaging work-up. We report the case of a 65-year-old man evaluated for a large multinodular goitre which caused compressive symptoms; the dominant nodule in the left lobe presented suspicious features on US. Thyroid function showed subclinical hypothyroidism, calcitonin was normal, serum thyroglobulin levels were low, and anti-thyroid antibodies were absent. The prevalent left nodule showed an intense uptake on 18F-FDG PET/CT but proved benign at FNAC. On the basis of the suspicious clinical and imaging features, total thyroidectomy was performed. Histology revealed a tall-cell variant of PTC with scattered expression of Tg and diffuse high expression of cytokeratin (CK) 19; RAI therapy was performed. Within 6 years of surgery, left laterocervical lymph-node recurrence was twice detected (first at levels II and III, then at levels IV and VI) by US and 18F-FDG-PET/CT and was confirmed by FNAC. Tg levels in the washout fluid proved clearly diagnostic of metastasis only in the second, larger, recurrence, whereas serum Tg levels (in the absence of anti-Tg antibodies) always remained undetectable on L-thyroxine therapy. Surgery was performed on both recurrences, and histology confirmed lymph-node metastasis of PTC. Immunohistochemical expression of Tg and CK 19 was similar to that of the primary tumour. No further relapses have occurred to date. Posttherapy (surgery and RAI) unstimulated serum Tg levels may not be a reliable marker of nodal recurrence in patients with differentiated thyroid cancer (DTC) that produces low amounts of Tg.
Copyright © 2020 Luca Foppiani et al.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures




References
-
- Sakorafas G. H., Koureas A., Mpampali I., Balalis D., Nasikas D., Ganztzoulas S. Patterns of lymph node metastasis in differentiated thyroid cancer; clinical implications with particular emphasis on the emerging role of compartment-oriented lymph node dissection. Oncology Research and Treatment. 2019;42(3):143–147. doi: 10.1159/000488905. - DOI - PubMed
-
- Haugen B. R., Alexander E. K., Bible K. C., et al. 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association Guidelines Task Force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26(1):1–133. doi: 10.1089/thy.2015.0020. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

