Stochasticity in Single-Entity Electrochemistry
- PMID: 33102927
- PMCID: PMC7584144
- DOI: 10.1016/j.coelec.2020.08.014
Stochasticity in Single-Entity Electrochemistry
Abstract
Most electrochemical processes are stochastic and discrete in nature. Yet experimental observables, e.g., i vs E, are typically smooth and deterministic, due to many events/processes, e.g., electron transfers, being averaged together. However, when the number of entities measured approaches a few or even one, stochasticity frequently emerges. Yet all is not lost! Probabilistic and statistical interpretation can generate insights matching or superseding those from macroscale/ensemble measurements, revealing phenomena that were hitherto averaged over. Herein, we review recent literature examples of stochastic processes in single-entity electrochemistry, highlighting strategies for interpreting stochasticity, contrasting them with macroscale measurements, and describing the insights generated.
Keywords: Brownian motion; Randomness; activated processes; probability; statistics.
Conflict of interest statement
Declaration of interests The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
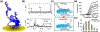
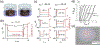


References
-
- Moazzenzade T, Huskens J, Lemay SG. Stochastic electrochemistry at ultralow concentrations: the case for digital sensors. Analyst 2020; 145:750–758. - PubMed
-
- Oja SM, Fan Y, Armstrong CM et al. Nanoscale Electrochemistry Revisited. Anal Chem 2016; 88:414–430. - PubMed
-
- Bentley CL, Kang M, Unwin PR. Nanoscale Surface Structure-Activity in Electrochemistry and Electrocatalysis. J Am Chem Soc 2019; 141:2179–2193. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
