Efficacy and Safety of ProGlide Versus Prostar XL Vascular Closure Devices in Transcatheter Aortic Valve Replacement: The RISPEVA Registry
- PMID: 33103545
- PMCID: PMC7763424
- DOI: 10.1161/JAHA.120.018042
Efficacy and Safety of ProGlide Versus Prostar XL Vascular Closure Devices in Transcatheter Aortic Valve Replacement: The RISPEVA Registry
Abstract
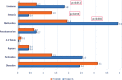
Background Transcatheter aortic valve replacement (TAVR) requires large-bore access, which is associated with bleeding and vascular complications. ProGlide and Prostar XL are vascular closure devices widely used in clinical practice, but their comparative efficacy and safety in TAVR is a subject of debate, owing to conflicting results among published studies. We aimed to compare outcomes with Proglide versus Prostar XL vascular closure devices after TAVR. Methods and Results This large-scale analysis was conducted using RISPEVA, a multicenter national prospective database of patients undergoing transfemoral TAVR treated with ProGlide versus Prostar XL vascular closure devices. Both multivariate and propensity score adjustments were performed. A total of 2583 patients were selected. Among them, 1361 received ProGlide and 1222 Prostar XL. The predefined primary end point was a composite of cardiovascular mortality, bleeding, and vascular complications assessed at 30 days and 1-year follow-up. At 30 days, there was a significantly greater reduction of the primary end point with ProGlide versus Prostar XL (13.8% versus 20.5%, respectively; multivariate adjusted odds ratio, 0.80 [95% CI, 0.65-0.99]; P=0.043), driven by a reduction of bleeding complications (9.1% versus 11.7%, respectively; multivariate adjusted odds ratio, 0.76 [95% CI, 0.58-0.98]; P=0.046). Propensity score analysis confirmed the significant reduction of major adverse cardiovascular events and bleeding risk with ProGlide. No significant differences in the primary end point were found between the 2 vascular closure devices at 1 year of follow-up (multivariate adjusted hazard ratio, 0.88 [95% CI, 0.72-1.10]; P=0.902). Comparable results were obtained by propensity score analysis. During the procedure, compared with Prostar XL, ProGlide yielded significant higher device success (99.2% versus 97.5%, respectively; P=0.001). Conclusions ProGlide has superior efficacy as compared with Prostar XL in TAVR procedures and is associated with a greater reduction of composite adverse events at short-term, driven by lower bleeding complications. Registration Information URL: clinicaltrials.gov; Unique identifier: NCT02713932.
Keywords: ProGlide; Prostar; transcatheter aortic valve replacement; vascular closure devices.
Conflict of interest statement
Dr Berti was a proctor for Abbott. Dr Navarese reports consulting fees/honoraria from Abbott, Astra‐Zeneca, Amgen, Bayer, and Sanofi‐Aventis; and grants from Abbott, and Amgen, outside the submitted work. Dr Giordano was a proctor for Abbott. The remaining authors have no disclosures to report.
Figures



References
-
- Borz B, Durand E, Godin M, Tron C, Canville A, Litzler PY, Bessou JP, Cribier A, Eltchaninoff H. Incidence, predictors and impact of bleeding after transcatheter aortic valve implantation using the balloon‐expandable Edwards prosthesis. Heart. 2013;860–865. - PubMed
-
- Sardar MR, Goldsweig AM, Abbott JD, Sharaf BL, Gordon PC, Ehsan A, Aronow HD. Vascular complications associated with transcatheter aortic valve replacement. Vasc Med. 2017;234–244. - PubMed
-
- Bavaria JE, Tommaso CL, Brindis RG, Carroll JD, Deeb GM, Feldman TE, Gleason TG, Horlick EM, Kavinsky CJ, Kumbhani DJ, et al. 2018 AATS/ACC/SCAI/STS expert consensus systems of care document: operator and institutional recommendations and requirements for transcatheter aortic valve replacement: a joint report of the American Association for Thoracic Surgery, American College of Cardiology, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2019;340–374. - PubMed
-
- Biancari F, Romppanen H, Savontaus M, Siljander A, Makikallio T, Piira OP, Piuhola J, Vilkki V, Ylitalo A, Vasankari T, et al. MANTA versus ProGlide vascular closure devices in transfemoral transcatheter aortic valve implantation. Int J Cardiol. 2018;29–31. - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical

