Humanin selectively prevents the activation of pro-apoptotic protein BID by sequestering it into fibers
- PMID: 33106313
- PMCID: PMC11843584
- DOI: 10.1074/jbc.RA120.013023
Humanin selectively prevents the activation of pro-apoptotic protein BID by sequestering it into fibers
Abstract
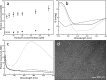
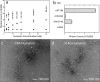
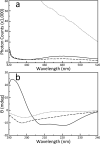
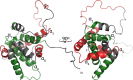
Members of the B-cell lymphoma (BCL-2) protein family regulate mitochondrial outer membrane permeabilization (MOMP), a phenomenon in which mitochondria become porous and release death-propagating complexes during the early stages of apoptosis. Pro-apoptotic BCL-2 proteins oligomerize at the mitochondrial outer membrane during MOMP, inducing pore formation. Of current interest are endogenous factors that can inhibit pro-apoptotic BCL-2 mitochondrial outer membrane translocation and oligomerization. A mitochondrial-derived peptide, Humanin (HN), was reported being expressed from an alternate ORF in the mitochondrial genome and inhibiting apoptosis through interactions with the pro-apoptotic BCL-2 proteins. Specifically, it is known to complex with BAX and BID. We recently reported the fibrillation of HN and BAX into β-sheets. Here, we detail the fibrillation between HN and BID. These fibers were characterized using several spectroscopic techniques, protease fragmentation with mass analysis, and EM. Enhanced fibrillation rates were detected with rising temperatures or pH values and the presence of a detergent. BID fibers are similar to those produced using BAX; however, the structures differ in final conformations of the BCL-2 proteins. BID fibers display both types of secondary structure in the fiber, whereas BAX was converted entirely to β-sheets. The data show that two distinct segments of BID are incorporated into the fiber structure, whereas other portions of BID remain solvent-exposed and retain helical structure. Similar analyses show that anti-apoptotic BCL-xL does not form fibers with humanin. These results support a general mechanism of sequestration of pro-apoptotic BCL-2 proteins into fibers by HN to inhibit MOMP.
Keywords: B-cell lymphoma 2 (Bcl-2) family; BID; amyloid; apoptosis; conformational change; electron microscopy (EM); fibers; humanin; β-sheet.
Conflict of interest statement
Conflict of interest— The authors declare that they have no conflicts of interest with the contents of this article.
Figures

Similar articles
-
Humanin induces conformational changes in the apoptosis regulator BAX and sequesters it into fibers, preventing mitochondrial outer-membrane permeabilization.J Biol Chem. 2019 Dec 13;294(50):19055-19065. doi: 10.1074/jbc.RA119.011297. Epub 2019 Nov 5. J Biol Chem. 2019. PMID: 31690630 Free PMC article.
-
BH3 domains other than Bim and Bid can directly activate Bax/Bak.J Biol Chem. 2011 Jan 7;286(1):491-501. doi: 10.1074/jbc.M110.167148. Epub 2010 Nov 1. J Biol Chem. 2011. PMID: 21041309 Free PMC article.
-
Examining BCL-2 family function with large unilamellar vesicles.J Vis Exp. 2012 Oct 5;(68):4291. doi: 10.3791/4291. J Vis Exp. 2012. PMID: 23070252 Free PMC article.
-
Discoveries and controversies in BCL-2 protein-mediated apoptosis.FEBS J. 2016 Jul;283(14):2690-700. doi: 10.1111/febs.13527. Epub 2015 Oct 27. FEBS J. 2016. PMID: 26411300 Review.
-
Bid: a Bax-like BH3 protein.Oncogene. 2008 Dec;27 Suppl 1:S93-104. doi: 10.1038/onc.2009.47. Oncogene. 2008. PMID: 19641510 Review.
Cited by
-
Inducible fold-switching as a mechanism to fibrillate pro-apoptotic BCL-2 proteins.Biopolymers. 2021 Oct;112(10):e23424. doi: 10.1002/bip.23424. Epub 2021 Mar 25. Biopolymers. 2021. PMID: 33764501 Free PMC article.
-
The Molecular Structure and Role of Humanin in Neural and Skeletal Diseases, and in Tissue Regeneration.Front Cell Dev Biol. 2022 Mar 16;10:823354. doi: 10.3389/fcell.2022.823354. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35372353 Free PMC article. Review.
-
Too much death can kill you: inhibiting intrinsic apoptosis to treat disease.EMBO J. 2021 Jul 15;40(14):e107341. doi: 10.15252/embj.2020107341. Epub 2021 May 26. EMBO J. 2021. PMID: 34037273 Free PMC article. Review.
-
Neuroprotective Action of Humanin and Humanin Analogues: Research Findings and Perspectives.Biology (Basel). 2023 Dec 16;12(12):1534. doi: 10.3390/biology12121534. Biology (Basel). 2023. PMID: 38132360 Free PMC article. Review.
-
Rhinovirus C causes heterogeneous infection and gene expression in airway epithelial cell subsets.Mucosal Immunol. 2023 Aug;16(4):386-398. doi: 10.1016/j.mucimm.2023.01.008. Epub 2023 Feb 14. Mucosal Immunol. 2023. PMID: 36796588 Free PMC article.
References
-
- Edlich F. BCL-2 proteins and apoptosis: recent insights and unknowns. Biochem. Biophys. Res. Commun. 2018;500:26–34. 10.1016/j.bbrc.2017.06.190 28676391. - DOI - PubMed
-
- Kale J., Osterlund E.J., Andrews D.W. BCL-2 family proteins: changing partners in the dance towards death. Cell Death Differ. 2018;25:65–80. 10.1038/cdd.2017.186 29149100. - DOI - PMC - PubMed
-
- Zhang M., Zheng J., Nussinov R., Ma B. Release of cytochrome c from Bax pores at the mitochondrial membrane. Sci. Rep. 2017;7 10.1038/s41598-017-02825-7 28572603. - DOI - PMC - PubMed
-
- Cosentino K., García-Sáez A.J. Bax and Bak pores: are we closing the circle? Trends Cell Biol. 2017;27:266–275. 10.1016/j.tcb.2016.11.004 27932064. - DOI - PMC - PubMed
-
- Cosentino K., García-Sáez A.J. MIM through MOM: the awakening of Bax and Bak pores. EMBO J. 2018;37 10.15252/embj.2018100340 30135068. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

