Bioadhesion in the oral cavity and approaches for biofilm management by surface modifications
- PMID: 33111157
- PMCID: PMC7666681
- DOI: 10.1007/s00784-020-03646-1
Bioadhesion in the oral cavity and approaches for biofilm management by surface modifications
Abstract
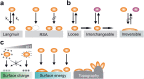
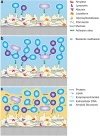
Background: All soft and solid surface structures in the oral cavity are covered by the acquired pellicle followed by bacterial colonization. This applies for natural structures as well as for restorative or prosthetic materials; the adherent bacterial biofilm is associated among others with the development of caries, periodontal diseases, peri-implantitis, or denture-associated stomatitis. Accordingly, there is a considerable demand for novel materials and coatings that limit and modulate bacterial attachment and/or propagation of microorganisms.
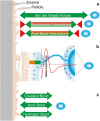
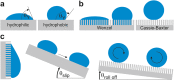
Objectives and findings: The present paper depicts the current knowledge on the impact of different physicochemical surface characteristics on bioadsorption in the oral cavity. Furthermore, it was carved out which strategies were developed in dental research and general surface science to inhibit bacterial colonization and to delay biofilm formation by low-fouling or "easy-to-clean" surfaces. These include the modulation of physicochemical properties such as periodic topographies, roughness, surface free energy, or hardness. In recent years, a large emphasis was laid on micro- and nanostructured surfaces and on liquid repellent superhydrophic as well as superhydrophilic interfaces. Materials incorporating mobile or bound nanoparticles promoting bacteriostatic or bacteriotoxic properties were also used. Recently, chemically textured interfaces gained increasing interest and could represent promising solutions for innovative antibioadhesion interfaces. Due to the unique conditions in the oral cavity, mainly in vivo or in situ studies were considered in the review.
Conclusion: Despite many promising approaches for modulation of biofilm formation in the oral cavity, the ubiquitous phenomenon of bioadsorption and adhesion pellicle formation in the challenging oral milieu masks surface properties and therewith hampers low-fouling strategies.
Clinical relevance: Improved dental materials and surface coatings with easy-to-clean properties have the potential to improve oral health, but extensive and systematic research is required in this field to develop biocompatible and effective substances.
Keywords: Biofilm management; Low-fouling surfaces; Nanostructured surfaces; Oral biofilms; Pellicle; Textured surfaces.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures


References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

