ESCargo: a regulatable fluorescent secretory cargo for diverse model organisms
- PMID: 33112725
- PMCID: PMC7927198
- DOI: 10.1091/mbc.E20-09-0591
ESCargo: a regulatable fluorescent secretory cargo for diverse model organisms
Abstract
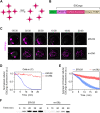
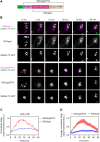
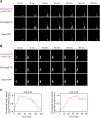
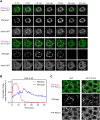
Membrane traffic can be studied by imaging a cargo protein as it transits the secretory pathway. The best tools for this purpose initially block export of the secretory cargo from the endoplasmic reticulum (ER) and then release the block to generate a cargo wave. However, previously developed regulatable secretory cargoes are often tricky to use or specific for a single model organism. To overcome these hurdles for budding yeast, we recently optimized an artificial fluorescent secretory protein that exits the ER with the aid of the Erv29 cargo receptor, which is homologous to mammalian Surf4. The fluorescent secretory protein forms aggregates in the ER lumen and can be rapidly disaggregated by addition of a ligand to generate a nearly synchronized cargo wave. Here we term this regulatable secretory protein ESCargo (Erv29/Surf4-dependent secretory cargo) and demonstrate its utility not only in yeast cells, but also in cultured mammalian cells, Drosophila cells, and the ciliate Tetrahymena thermophila. Kinetic studies indicate that rapid export from the ER requires recognition by Erv29/Surf4. By choosing an appropriate ER signal sequence and expression vector, this simple technology can likely be used with many model organisms.
Figures
Similar articles
-
Visualizing Secretory Cargo Transport in Budding Yeast.Curr Protoc Cell Biol. 2019 Jun;83(1):e80. doi: 10.1002/cpcb.80. Epub 2018 Nov 10. Curr Protoc Cell Biol. 2019. PMID: 30414385 Free PMC article.
-
Dual Independent Roles of the p24 Complex in Selectivity of Secretory Cargo Export from the Endoplasmic Reticulum.Cells. 2020 May 22;9(5):1295. doi: 10.3390/cells9051295. Cells. 2020. PMID: 32456004 Free PMC article.
-
Export of polybasic motif-containing secretory proteins BMP8A and SFRP1 from the endoplasmic reticulum is regulated by surfeit locus protein 4.J Biol Chem. 2022 Dec;298(12):102687. doi: 10.1016/j.jbc.2022.102687. Epub 2022 Nov 9. J Biol Chem. 2022. PMID: 36370847 Free PMC article.
-
Cargo loading at the ER.Mol Membr Biol. 2010 Nov;27(8):398-411. doi: 10.3109/09687688.2010.506203. Mol Membr Biol. 2010. PMID: 21142873 Review.
-
Receptor-mediated protein transport in the early secretory pathway.Trends Biochem Sci. 2007 Aug;32(8):381-8. doi: 10.1016/j.tibs.2007.06.006. Epub 2007 Jul 6. Trends Biochem Sci. 2007. PMID: 17618120 Review.
Cited by
-
Genetically encoded fluorescent tools: Shining a little light on ER-to-Golgi transport.Free Radic Biol Med. 2022 Apr;183:14-24. doi: 10.1016/j.freeradbiomed.2022.03.004. Epub 2022 Mar 8. Free Radic Biol Med. 2022. PMID: 35272000 Free PMC article. Review.
-
SURF4-induced tubular ERGIC selectively expedites ER-to-Golgi transport.Dev Cell. 2022 Feb 28;57(4):512-525.e8. doi: 10.1016/j.devcel.2021.12.018. Epub 2022 Jan 19. Dev Cell. 2022. PMID: 35051356 Free PMC article.
-
CRISPR/Cas9-mediated point mutations improve α-amylase secretion in Saccharomyces cerevisiae.FEMS Yeast Res. 2022 Jul 15;22(1):foac033. doi: 10.1093/femsyr/foac033. FEMS Yeast Res. 2022. PMID: 35776981 Free PMC article.
-
The secretory pathway in Tetrahymena is organized for efficient constitutive secretion at ciliary pockets.iScience. 2024 Oct 9;27(11):111123. doi: 10.1016/j.isci.2024.111123. eCollection 2024 Nov 15. iScience. 2024. PMID: 39498308 Free PMC article.
-
Bioorthogonal, Fluorogenic Targeting of Voltage-Sensitive Fluorophores for Visualizing Membrane Potential Dynamics in Cellular Organelles.J Am Chem Soc. 2022 Jul 13;144(27):12138-12146. doi: 10.1021/jacs.2c02664. Epub 2022 Jul 1. J Am Chem Soc. 2022. PMID: 35776693 Free PMC article.
References
-
- Barlowe C, Helenius A (2016). Cargo capture and bulk flow in the early secretory pathway. Annu Rev Cell Dev Biol 32, 197–222. - PubMed
-
- Bevis BJ, Hammond AT, Reinke CA, Glick BS (2002). De novo formation of transitional ER sites and Golgi structures in Pichia pastoris . Nat Cell Biol 4, 750–756. - PubMed
-
- Boncompain G, Divoux S, Gareil N, de Forges H, Lescure A, Latreche L, Mercanti V, Jollivet F, Raposo G, Perez F (2012). Synchronization of secretory protein traffic in populations of cells. Nat Methods 9, 493–498. - PubMed
-
- Boncompain G, Perez F (2013). Fluorescence-based analysis of trafficking in mammalian cells. Methods Cell Biol 118, 179–194. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

