Kynurenine Promotes RANKL-Induced Osteoclastogenesis In Vitro by Activating the Aryl Hydrocarbon Receptor Pathway
- PMID: 33114603
- PMCID: PMC7662708
- DOI: 10.3390/ijms21217931
Kynurenine Promotes RANKL-Induced Osteoclastogenesis In Vitro by Activating the Aryl Hydrocarbon Receptor Pathway
Abstract
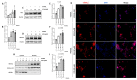
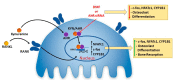
There is increasing evidence of the involvement of the tryptophan metabolite kynurenine (KYN) in disrupting osteogenesis and contributing to aging-related bone loss. Here, we show that KYN has an effect on bone resorption by increasing osteoclastogenesis. We have previously reported that in vivo treatment with KYN significantly increased osteoclast number lining bone surfaces. Here, we report the direct effect of KYN on receptor activator of nuclear factor kappa-B ligand (RANKL)-induced osteoclastogenesis in Raw 264.7 macrophage cells, and we propose a potential mechanism for these KYN-mediated effects. We show that KYN/RANKL treatment results in enhancement of RANKL-induced osteoclast differentiation. KYN drives upregulation and activation of the key osteoclast transcription factors, c-fos and NFATc1 resulting in an increase in the number of multinucleated TRAP+ osteoclasts, and in hydroxyapatite bone resorptive activity. Mechanistically, the KYN receptor, aryl hydrocarbon receptor (AhR), plays an important role in the induction of osteoclastogenesis. We show that blocking AhR signaling using an AhR antagonist, or AhR siRNA, downregulates the KYN/RANKL-mediated increase in c-fos and NFATc1 and inhibits the formation of multinucleated TRAP + osteoclasts. Altogether, this work highlights that the novelty of the KYN and AhR pathways might have a potential role in helping to regulate osteoclast function with age and supports pursuing additional research to determine if they are potential therapeutic targets for the prevention or treatment of osteoporosis.
Keywords: AhR; Kynurenine; NFATc1; c-fos; osteoclast.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures



References
-
- Cao J.J., Wronski T.J., Iwaniec U., Phleger L., Kurimoto P., Boudignon B., Halloran B.P. Aging increases stromal/osteoblastic cell-induced osteoclastogenesis and alters the osteoclast precursor pool in the mouse. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2005;20:1659–1668. doi: 10.1359/JBMR.050503. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

