Gypenosides improves nonalcoholic fatty liver disease induced by high-fat diet induced through regulating LPS/TLR4 signaling pathway
- PMID: 33121337
- PMCID: PMC7714522
- DOI: 10.1080/15384101.2020.1829800
Gypenosides improves nonalcoholic fatty liver disease induced by high-fat diet induced through regulating LPS/TLR4 signaling pathway
Abstract
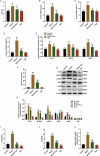
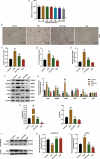
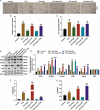
Background The contents of lipopolysaccharide (LPS) and Toll-like receptor 4 (TLR4) are significantly increased during the progression of nonalcoholic fatty liver disease (NAFLD). The study investigated the role of the LPS/TLR4 signaling pathway in improving gypenosides (Gyp) on NAFLD. Methods NAFLD model were established in rats and treated by Gyp. Pathological changes of liver tissues were observed by Hematoxylin and Eosin (HE) staining. Lipid metabolism and insulin resistance were measured. Expressions of inflammatory factors and protein of LPS/TLR4 downstream pathway were detected by qRT-PCR and Western blotting. THLE-2 cells were treated by free-fatty acid (FFA), Gyp, and LPS, and then transfected with TLR4. Next, cell viability was detected by MTT. Lipid droplet deposition and Triglyceride (TG) content were determined by Oil Red O staining and ELISA. Results Gyp protected fatty liver tissues in NAFLD model, and significantly reversed cholesterol increased by high-fat diet. Moreover, Gyp increased SOD content and decreased the contents of AST, ALT, MDA, HSI, FBG, FINS, HOMA-IR, IL-1β, and TNF-α, and promoted the expressions of TLR4, LPS, MyD88, p-IκBα, and reduced the expressions of p-p65 and IκBα in the NAFLD model. Gyp treatment significantly reduced lipid droplet deposition, increased TG content and MyD88, p-IκBα, p-p65 in FFA-induced liver cells, but LPS and TLR4 greatly reversed improvement of FFA by Gyp. Conclusion Gypenosides could improve liver function, lipid metabolism, insulin resistance, and levels of inflammatory factors in NAFLD model by regulating LPS/TLR4 signaling pathway in vitro and in vivo.
Keywords: Gypenosides; LPS/TLR4 signaling pathway; insulin resistance; lipid metabolism; nonalcoholic fatty liver disease.
Conflict of interest statement
No potential conflict of interest was reported by the authors.
Figures
References
-
- Chakravarthy MV, Waddell T, Banerjee R, et al. Nutrition and nonalcoholic fatty liver disease: current perspectives. Gastroenterol Clin North Am. 2020;49(1):63–94. - PubMed
-
- Tang XR, Wang JX, Fu L, et al. Effects of total flavonoids in astragali complanati semen on liver lipid level and ERalpha expression on liver in hyperlipidemia rats with kidney-Yang deficiency pattern. Zhongguo Zhong Yao Za Zhi. 2018;43:2365–2371. - PubMed
-
- Kiziltas S, Ata P, Colak Y, et al. TLR4 gene polymorphism in patients with nonalcoholic fatty liver disease in comparison to healthy controls. Metab Syndr Relat Disord. 2014;12(3):165–170. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
