Auxiliary diagnostic value of tumor biomarkers in pleural fluid for lung cancer-associated malignant pleural effusion
- PMID: 33121490
- PMCID: PMC7596935
- DOI: 10.1186/s12931-020-01557-z
Auxiliary diagnostic value of tumor biomarkers in pleural fluid for lung cancer-associated malignant pleural effusion
Abstract
Background: Pleural effusion (PE) can be divided into benign pleural effusion (BPE) and malignant pleural effusion (MPE). There is no consensus on the identification of lung cancer-associated MPE using the optimal cut-off levels from five common tumor biomarkers (CEA, CYFRA 21-1, CA125, SCC-Ag, and NSE). Therefore, we aimed to find indicators for the auxiliary diagnosis of lung cancer-associated MPE by analyzing and then validating the optimal threshold levels of these biomarkers in pleural fluid (PF) and serum, as well as the PF/serum ratio.
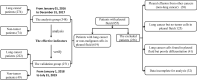
Patients and method: The study has two sets of patients, i.e. the training set and the test set. In the training set, 348 patients with PE, between January 1, 2016 and December 31, 2017, were divided into BPE and MPE based on the cytological diagnosis. Subsequently, the optimal cut-off levels of tumor biomarkers were analyzed. In the test set, the diagnostic compliance rate was verified with 271 patients with PE from January 1, 2018 to July 31, 2019 to evaluate the auxiliary diagnostic value of the aforementioned indicators.
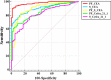
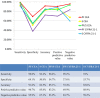
Result: In the training set, PF CEA at the cut-off value of 5.23 ng/ml was the most effective indicator for MPE compared with other tumor biomarkers (all p < 0.001). In the test set, PF CEA at the cut-off value of 5.23 ng/ml showed the highest sensitivity, specificity and accuracy, positive and negative predictive value among other tumor biomarkers, which were 99.0%, 69.1%, 91.6%, 90.7%, and 95.9%, respectively.
Conclusion: PF CEA at the cut-off level of 5.23 ng/ml was the most effective indicator for identifying lung cancer-associated MPE among the five common tumor biomarkers.
Keywords: CEA; Lung cancer; Pleural effusion; Serum; Tumor markers.
Conflict of interest statement
The authors state that they have no conflict of interests.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous