Fibroblast-like cells change gene expression of bone remodelling markers in transwell cultures
- PMID: 33121539
- PMCID: PMC7596965
- DOI: 10.1186/s40001-020-00453-y
Fibroblast-like cells change gene expression of bone remodelling markers in transwell cultures
Abstract
Introduction: Periprosthetic fibroblast-like cells (PPFs) play an important role in aseptic loosening of arthroplasties. Various studies have examined PPF behavior in monolayer culture systems. However, the periprosthetic tissue is a three-dimensional (3D) mesh, which allows the cells to interact in a multidirectional way. The expression of bone remodeling markers of fibroblast-like cells in a multilayer environment changes significantly versus monolayer cultures without the addition of particles or cytokine stimulation. Gene expression of bone remodeling markers was therefore compared in fibroblast-like cells from different origins and dermal fibroblasts under transwell culture conditions versus monolayer cultures.
Methods: PPFs from periprosthetic tissues (n = 12), osteoarthritic (OA) synovial fibroblast-like cells (SFs) (n = 6), and dermal fibroblasts (DFs) were cultured in monolayer (density 5.5 × 103/cm2) or multilayer cultures (density 8.5 × 105/cm2) for 10 or 21 days. Cultures were examined via histology, TRAP staining, immunohistochemistry (anti-S100a4), and quantitative real-time PCR.
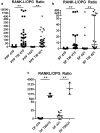
Results: Fibroblast-like cells (PPFs/SFs) and dermal fibroblasts significantly increased the expression of RANKL and significantly decreased the expression of ALP, COL1A1, and OPG in multilayer cultures. PPFs and SFs in multilayer cultures further showed a higher expression of cathepsin K, MMP-13, and TNF-α. In multilayer PPF cultures, the mRNA level of TRAP was also found to be significantly increased.
Conclusion: The multilayer cultures are able to induce significant expression changes in fibroblast-like cells depending on the nature of cellular origin without the addition of any further stimulus. This system might be a useful tool to get more in vivo like results regarding fibroblast-like cell cultures.
Keywords: Aseptic loosening; Bone remodeling; Fibroblast-like cells; Implant material; Transwell culture.
Conflict of interest statement
The authors indicate no potential conflict of interests.
Figures




Similar articles
-
Impact of Periprosthetic Fibroblast-Like Cells on Osteoclastogenesis in Co-Culture with Peripheral Blood Mononuclear Cells Varies Depending on Culture System.Int J Mol Sci. 2019 May 26;20(10):2583. doi: 10.3390/ijms20102583. Int J Mol Sci. 2019. PMID: 31130703 Free PMC article.
-
Comparison of cathepsins K and S expression within the rheumatoid and osteoarthritic synovium.Arthritis Rheum. 2002 Mar;46(3):663-74. doi: 10.1002/art.10114. Arthritis Rheum. 2002. PMID: 11920402
-
Factors regulating bone remodeling processes in aseptic implant loosening.J Orthop Res. 2017 Feb;35(2):248-257. doi: 10.1002/jor.23274. Epub 2016 May 8. J Orthop Res. 2017. PMID: 27116254
-
Naringin increases osteoprotegerin expression in fibroblasts from periprosthetic membrane by the Wnt/β-catenin signaling pathway.J Orthop Surg Res. 2020 Dec 10;15(1):600. doi: 10.1186/s13018-020-02145-z. J Orthop Surg Res. 2020. PMID: 33302980 Free PMC article.
-
Understanding Fibroblast Behavior in 3D Biomaterials.Tissue Eng Part B Rev. 2022 Jun;28(3):569-578. doi: 10.1089/ten.TEB.2021.0010. Epub 2021 Aug 16. Tissue Eng Part B Rev. 2022. PMID: 34102862 Review.
Cited by
-
Impact of Periprosthetic Fibroblast-Like Cells on Osteoclastogenesis in Co-Culture with Peripheral Blood Mononuclear Cells Varies Depending on Culture System.Int J Mol Sci. 2019 May 26;20(10):2583. doi: 10.3390/ijms20102583. Int J Mol Sci. 2019. PMID: 31130703 Free PMC article.
-
Dental and Orthopaedic Implant Loosening: Overlap in Gene Expression Regulation.Front Immunol. 2022 Feb 11;13:820843. doi: 10.3389/fimmu.2022.820843. eCollection 2022. Front Immunol. 2022. PMID: 35222398 Free PMC article.
-
Exosome: Function and Application in Inflammatory Bone Diseases.Oxid Med Cell Longev. 2021 Aug 31;2021:6324912. doi: 10.1155/2021/6324912. eCollection 2021. Oxid Med Cell Longev. 2021. Retraction in: Oxid Med Cell Longev. 2024 Jan 9;2024:9806854. doi: 10.1155/2024/9806854. PMID: 34504641 Free PMC article. Retracted. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

