Characteristics of clinical trials evaluating cardiovascular therapies for Coronavirus Disease 2019 Registered on ClinicalTrials.gov: a cross sectional analysis
- PMID: 33121978
- PMCID: PMC7586939
- DOI: 10.1016/j.ahj.2020.10.065
Characteristics of clinical trials evaluating cardiovascular therapies for Coronavirus Disease 2019 Registered on ClinicalTrials.gov: a cross sectional analysis
Abstract
Morbidity and mortality associated with COVID-19 has increased exponentially, and patients with cardiovascular (CV) disease are at risk for poor outcomes. Several lines of evidence suggest a potential role for CV therapies in COVID-19 treatment. Characteristics of clinical trials of CV therapies related to COVID-19 registered on ClinicalTrials.gov have not been described.
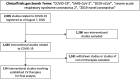
Methods: ClinicalTrials.gov was queried on August 7, 2020 for COVID-19 related trials. Studies evaluating established CV drugs, other fibrinolytics (defibrotide), and extracorporeal membrane oxygenation were included. Studies evaluating anti-microbial, convalescent plasma, non-colchicine anti-inflammatory, and other therapies were excluded. Trial characteristics were tabulated from study-specific entries.
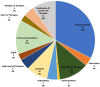
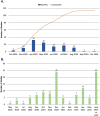
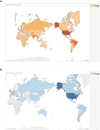
Results: A total of 2,935 studies related to COVID-19 were registered as of August 7, 2020. Of these, 1,645 were interventional studies, and the final analytic cohort consisted of 114 studies evaluating 10 CV therapeutic categories. Antithrombotics (32.5%; n = 37) were most commonly evaluated, followed by pulmonary vasodilators (14.0%; n = 16), renin-angiotensin-aldosterone system-related therapies (12.3%; n = 14), and colchicine (8.8%; n = 10). Trials evaluating multiple CV therapy categories and CV therapies in combination with non-CV therapies encompassed 4.4% (n = 5) and 9.6% (n = 11) of studies, respectively. Most studies were designed for randomized allocation (87.7%; n = 100), enrollment of less than 1000 participants (86.8%; n = 99), single site implementation (55.3%; n = 63), and had a primary outcome of mortality or a composite including mortality (56.1%; n = 64). Most study populations consisted of patients hospitalized with COVID-19 (81.6%; n = 93). At the time of database query, 28.9% (n = 33) of studies were not yet recruiting and the majority were estimated to be completed after December 2020 (67.8%; n = 78). Most lead sponsors were located in North America (43.9%; n = 50) or Europe (36.0%; n = 41).
Conclusions: A minority (7%) of clinical trials related to COVID-19 registered on ClinicalTrials.gov plan to evaluate CV therapies. Of CV therapy studies, most were planned to be single center, enroll less than 1000 inpatients, sponsored by European or North American academic institutions, and estimated to complete after December 2020. Collectively, these findings underscore the need for a network of sites with a platform protocol for rapid evaluation of multiple therapies and generalizability to inform clinical care and health policy for COVID-19 moving forward.
Copyright © 2020 Elsevier Inc. All rights reserved.
Figures
References
-
- COVID-19 situation reports [Internet]. [cited 2020 May 3]. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situatio....
-
- RECOVERY Collaborative Group. Horby P., Lim W.S., et al. Dexamethasone in hospitalized patients with COVID-19—Preliminary Report. N Engl J Med. 2020 Jul 17 - PubMed
-
- Beigel J.H., Tomashek K.M., Dodd L.E., et al. Remdesivir for the treatment of COVID-19—preliminary report. N Engl J Med. 2020 May;22 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

