Long Non-Coding RNA USP2-AS1 Accelerates Cell Proliferation and Migration in Ovarian Cancer by Sponging miR-520d-3p and Up-Regulating KIAA1522
- PMID: 33122952
- PMCID: PMC7591011
- DOI: 10.2147/CMAR.S268863
Long Non-Coding RNA USP2-AS1 Accelerates Cell Proliferation and Migration in Ovarian Cancer by Sponging miR-520d-3p and Up-Regulating KIAA1522
Abstract
Background: Ovarian cancer is one of the malignant tumors attacking the female reproductive system. Currently, increasing studies have clearly determined the importance of long non-coding RNAs (lncRNAs) in various human cancers including ovarian cancer. However, the role and in-depth mechanism of ubiquitin specific peptidase 2 antisense RNA 1 (USP2-AS1) in ovarian cancer have been not reported yet.
Purpose: We were absorbed into exploring the character of USP2-AS1 in ovarian cancer.
Methods: RT-qPCR analysis reflected gene expression. The GEPIA database provided further evidences, and bioinformatics tools analyzed the potential molecules downstream USP2-AS1 in ovarian cancer. The changes on ovarian cancer cellular functions were assessed via EdU, TUNEL, JC-1 and transwell assays. RNA pull down, RIP and luciferase reporter assays estimated molecule interactions.
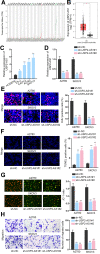
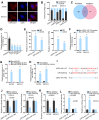
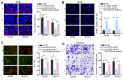
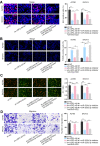
Results: USP2-AS1 was obviously up-regulated in ovarian cancer tissues and cell lines. Inhibiting USP2-AS1 had anti-proliferation, pro-apoptosis, and anti-migration effects on ovarian cancer cells. Furthermore, we confirmed that USP2-AS1 sequestered miR-520d-3p to enhance KIAA1522. In addition, miR-520d-3p silence reversed the effect of depleted USP2-AS1 on ovarian cancer cellular behaviors, while such reversion was then abolished by KIAA1522 knockdown.
Conclusion: USP2-AS1 facilitated ovarian cancer progression via miR-520d-3p/KIAA1522 axis, implying USP2-AS1 as a new perspective for the treatment of ovarian cancer.
Keywords: KIAA1522; USP2-AS1; miR-520d-3p; ovarian cancer.
© 2020 Guo et al.
Conflict of interest statement
The authors declare that they have no competing interests in this study.
Figures

Similar articles
-
Targeting the deubiquitinase USP2 for malignant tumor therapy (Review).Oncol Rep. 2023 Oct;50(4):176. doi: 10.3892/or.2023.8613. Epub 2023 Aug 18. Oncol Rep. 2023. PMID: 37594087 Free PMC article. Review.
-
LncRNA FGD5-AS1 accelerates cell proliferation in pancreatic cancer by regulating miR-520a-3p/KIAA1522 axis.Cancer Biol Ther. 2021 Mar 4;22(3):257-266. doi: 10.1080/15384047.2021.1883184. Epub 2021 Apr 2. Cancer Biol Ther. 2021. PMID: 33794727 Free PMC article.
-
MYBL2-induced PITPNA-AS1 upregulates SIK2 to exert oncogenic function in triple-negative breast cancer through miR-520d-5p and DDX54.J Transl Med. 2021 Aug 5;19(1):333. doi: 10.1186/s12967-021-02956-6. J Transl Med. 2021. PMID: 34353336 Free PMC article.
-
Down-regulation of ZNF252P-AS1 alleviates ovarian cancer progression by binding miR-324-3p to downregulate LY6K.J Ovarian Res. 2022 Jan 3;15(1):1. doi: 10.1186/s13048-021-00933-7. J Ovarian Res. 2022. PMID: 34980214 Free PMC article.
-
LncRNA FLVCR1-AS1 promotes proliferation, migration and activates Wnt/β-catenin pathway through miR-381-3p/CTNNB1 axis in breast cancer.Cancer Cell Int. 2020 Jun 5;20:214. doi: 10.1186/s12935-020-01247-2. eCollection 2020. Cancer Cell Int. 2020. PMID: 32518523 Free PMC article.
Cited by
-
Identification and Characterization of an Ageing-Associated 13-lncRNA Signature That Predicts Prognosis and Immunotherapy in Hepatocellular Carcinoma.J Oncol. 2023 Feb 17;2023:4615297. doi: 10.1155/2023/4615297. eCollection 2023. J Oncol. 2023. PMID: 36844873 Free PMC article.
-
lncHUB2: aggregated and inferred knowledge about human and mouse lncRNAs.Database (Oxford). 2023 Mar 4;2023:baad009. doi: 10.1093/database/baad009. Database (Oxford). 2023. PMID: 36869839 Free PMC article.
-
Targeting the deubiquitinase USP2 for malignant tumor therapy (Review).Oncol Rep. 2023 Oct;50(4):176. doi: 10.3892/or.2023.8613. Epub 2023 Aug 18. Oncol Rep. 2023. PMID: 37594087 Free PMC article. Review.
-
LncRNA USP2-AS1 facilitates the osteogenic differentiation of bone marrow mesenchymal stem cells by targeting KDM3A/ETS1/USP2 to activate the Wnt/β-catenin signaling pathway.RNA Biol. 2024 Jan;21(1):1-13. doi: 10.1080/15476286.2023.2290771. Epub 2023 Dec 22. RNA Biol. 2024. PMID: 38131611 Free PMC article.
-
Latest Update on lncRNA in Epithelial Ovarian Cancer-A Scoping Review.Cells. 2025 Apr 7;14(7):555. doi: 10.3390/cells14070555. Cells. 2025. PMID: 40214508 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

