Increased Plasma Heparanase Activity in COVID-19 Patients
- PMID: 33123154
- PMCID: PMC7573491
- DOI: 10.3389/fimmu.2020.575047
Increased Plasma Heparanase Activity in COVID-19 Patients
Abstract
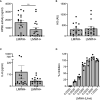
Reports suggest a role of endothelial dysfunction and loss of endothelial barrier function in COVID-19. It is well established that the endothelial glycocalyx-degrading enzyme heparanase contributes to vascular leakage and inflammation. Low molecular weight heparins (LMWH) serve as an inhibitor of heparanase. We hypothesize that heparanase contributes to the pathogenesis of COVID-19, and that heparanase may be inhibited by LMWH. To test this hypothesis, heparanase activity and heparan sulfate levels were measured in plasma of healthy controls (n = 10) and COVID-19 patients (n = 48). Plasma heparanase activity and heparan sulfate levels were significantly elevated in COVID-19 patients. Heparanase activity was associated with disease severity including the need for intensive care, lactate dehydrogenase levels, and creatinine levels. Use of prophylactic LMWH in non-ICU patients was associated with a reduced heparanase activity. Since there is no other clinically applied heparanase inhibitor currently available, therapeutic treatment of COVID-19 patients with low molecular weight heparins should be explored.
Keywords: COVID-19; LMWH (low molecular weight heparin); glycocalyx damage; heparanase; inflammation; vascular leakage.
Copyright © 2020 Buijsers, Yanginlar, de Nooijer, Grondman, Maciej-Hulme, Jonkman, Janssen, Rother, de Graaf, Pickkers, Kox, Joosten, Nijenhuis, Netea, Hilbrands, van de Veerdonk, Duivenvoorden, de Mast and van der Vlag.
Figures



References
-
- Li Z, Wu M, Yao J, Guo J, Liao X, Song S, et al. Caution on Kidney Dysfunctions of COVID-19 Patients. medRxiv (2020). 10.1101/2020.02.08.20021212 - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

