Cannabis sativa extracts protect LDL from Cu2+-mediated oxidation
- PMID: 33123676
- PMCID: PMC7592720
- DOI: 10.1186/s42238-020-00042-0
Cannabis sativa extracts protect LDL from Cu2+-mediated oxidation
Abstract
Background: Multiple therapeutic properties have been attributed to Cannabis sativa. However, further research is required to unveil the medicinal potential of Cannabis and the relationship between biological activity and chemical profile.
Objectives: The primary objective of this study was to characterize the chemical profile and antioxidant properties of three varieties of Cannabis sativa available in Uruguay during progressive stages of maturation.
Methods: Fresh samples of female inflorescences from three stable Cannabis sativa phenotypes, collected at different time points during the end of the flowering period were analyzed. Chemical characterization of chloroform extracts was performed by 1H-NMR. The antioxidant properties of the cannabis sativa extracts, and pure cannabinoids, were measured in a Cu2+-induced LDL oxidation assay.
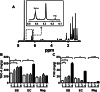
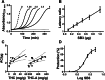
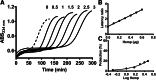
Results: The main cannabinoids in the youngest inflorescences were tetrahydrocannabinolic acid (THC-A, 242 ± 62 mg/g) and tetrahydrocannabinol (THC, 7.3 ± 6.5 mg/g). Cannabinoid levels increased more than twice in two of the mature samples. A third sample showed a lower and constant concentration of THC-A and THC (177 ± 25 and 1 ± 1, respectively). The THC-A/THC rich cannabis extracts increased the latency phase of LDL oxidation by a factor of 1.2-3.5 per μg, and slowed down the propagation phase of lipoperoxidation (IC50 1.7-4.6 μg/mL). Hemp, a cannabidiol (CBD, 198 mg/g) and cannabidiolic acid (CBD-A, 92 mg/g) rich variety, also prevented the formation of conjugated dienes during LDL oxidation. In fact, 1 μg of extract was able to stretch the latency phase 3.7 times and also to significantly reduce the steepness of the propagation phase (IC50 of 8 μg/mL). Synthetic THC lengthened the duration of the lag phase by a factor of 21 per μg, while for the propagation phase showed an IC50 ≤ 1 μg/mL. Conversely, THC-A was unable to improve any parameter. Meanwhile, the presence of 1 μg of pure CBD and CBD-A increased the initial latency phase 4.8 and 9.4 times, respectively, but did not have an effect on the propagation phase.
Conclusion: Cannabis whole extracts acted on both phases of lipid oxidation in copper challenged LDL. Those effects were just partially related with the content of cannabinoids and partially recapitulated by isolated pure cannabinoids. Our results support the potentially beneficial effects of cannabis sativa whole extracts on the initial phase of atherosclerosis.
Keywords: Cannabis sativa; atherosclerosis; hemp; low-density lipoprotein; maturation; oxidation; phytocannabinoids.
Conflict of interest statement
7.4.Competing interests The authors declare no conflicts of interest with the contents of this article.
Figures


References
-
- Chapman MJ, Goldstein S, Latencyrange D, Laplaud PM. A density gradient ultracentrifugal procedure for the isolation of the major lipoprotein classes from human serum. J Lipid Res. 1981;22:339–358. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

