Biomarker-driven therapies for previously treated squamous non-small-cell lung cancer (Lung-MAP SWOG S1400): a biomarker-driven master protocol
- PMID: 33125909
- PMCID: PMC8109255
- DOI: 10.1016/S1470-2045(20)30475-7
Biomarker-driven therapies for previously treated squamous non-small-cell lung cancer (Lung-MAP SWOG S1400): a biomarker-driven master protocol
Abstract
Background: The Lung Cancer Master Protocol (Lung-MAP; S1400) is a completed biomarker-driven master protocol designed to address an unmet need for better therapies for squamous non-small-cell lung cancer. Lung-MAP (S1400) was created to establish an infrastructure for biomarker screening and rapid regulatory intent evaluation of targeted therapies and was the first biomarker-driven master protocol initiated with the US National Cancer Institute (NCI).
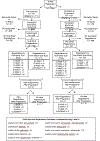
Methods: Lung-MAP (S1400) was done within the National Clinical Trials Network of the NCI using a public-private partnership. Eligible patients were aged 18 years or older, had stage IV or recurrent squamous non-small-cell lung cancer, had previously been treated with platinum-based chemotherapy, and had an Eastern Cooperative Oncology Group (ECOG) performance status of 0-2. The study included a screening component using the FoundationOne assay (Foundation Medicine, Cambridge, MA, USA) for next-generation sequencing, and a clinical trial component with biomarker-driven substudies and non-match substudies for patients who were ineligible for biomarker-driven substudies. Patients were pre-screened and received their substudy assignment upon progression, or they were screened at progression and received their substudy assignment upon completion of testing. Patients could enrol onto additional substudies after progression on a substudy. The study is registered with ClinicalTrials.gov, NCT02154490, and all research related to Lung-MAP (S1400) is completed.
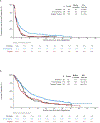
Findings: Between June 16, 2014, and Jan 28, 2019, 1864 patients enrolled and 1841 (98·9%) submitted tissue. 1674 (90·9%) of 1841 patients had biomarker results, and 1404 (83·9%) of 1674 patients received a substudy assignment. Of the assigned patients, 655 (46·7%) registered to a substudy. The biomarker-driven substudies evaluated taselisib (targeting PIK3CA alterations), palbociclib (cell cycle gene alterations), AZD4547 (FGFR alteration), rilotumumab plus erlotinib (MET), talazoparib (homologous recombination repair deficiency), and telisotuzumab vedotin (MET). The non-match substudies evaluated durvalumab, and nivolumab plus ipilimumab for anti-PD-1 or anti-PD-L1-naive disease, and durvalumab plus tremelimumab for anti-PD-1 or anti-PD-L1 relapsed disease. Combining data from the substudies, ten (7·0%) of 143 patients responded to targeted therapy, 53 (16·8%) of 315 patients responded to anti-PD-1 or anti-PD-L1 therapy for immunotherapy-naive disease, and three (5·4%) of 56 responded to docetaxel in the second line of therapy. Median overall survival was 5·9 months (95% CI 4·8-7·8) for the targeted therapy groups, 7·7 months (6·7-9·2) for the docetaxel groups, and 10·8 months (9·4-12·3) for the anti-PD-1 or anti-PD-L1-containing groups. Median progression-free survival was 2·5 months (95% CI 1·7-2·8) for the targeted therapy groups, 2·7 months (1·9-2·9) for the docetaxel groups, and 3·0 months (2·7-3·9) for the anti-PD-1 or anti-PD-L1-containing groups.
Interpretation: Lung-MAP (S1400) met its goal to quickly address biomarker-driven therapy questions in squamous non-small-cell lung cancer. In early 2019, a new screening protocol was implemented expanding to all histological types of non-small-cell lung cancer and to add focus on immunotherapy combinations for anti-PD-1 and anti-PD-L1 therapy-relapsed disease. With these changes, Lung-MAP continues to meet its goal to focus on unmet needs in the treatment of advanced lung cancers.
Funding: US National Institutes of Health, and AbbVie, Amgen, AstraZeneca, Bristol Myers Squibb, Genentech, and Pfizer through the Foundation for the National Institutes of Health.
Copyright © 2020 Elsevier Ltd. All rights reserved.
Figures
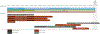
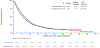
Comment in
-
The quest for efficient trial designs in precision oncology.Lancet Oncol. 2020 Dec;21(12):1539-1541. doi: 10.1016/S1470-2045(20)30553-2. Epub 2020 Oct 27. Lancet Oncol. 2020. PMID: 33125908 No abstract available.
-
Therapeutic utility of Lung-MAP: ushering into an era of genomic and biomarker-driven clinical trials.Signal Transduct Target Ther. 2021 Apr 1;6(1):141. doi: 10.1038/s41392-021-00557-9. Signal Transduct Target Ther. 2021. PMID: 33795635 Free PMC article. No abstract available.
References
-
- Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature 2018; 553: 446–54. - PubMed
-
- Roskoski R Jr, Properties of FDA-approved small molecule protein kinase inhibitors. Pharmacol Res 2019; 144: 19–50. - PubMed
-
- Morgensztern D, Waqar S, Subramanian J, Gao F, Govindan R. Improving survival for stage IV non-small cell lung cancer: a surveillance, epidemiology, and end results survey from 1990 to 2005. J Thorac Oncol 2009; 4: 1524–9. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- P30 CA093373/CA/NCI NIH HHS/United States
- UG1 CA189971/CA/NCI NIH HHS/United States
- UL1 TR001863/TR/NCATS NIH HHS/United States
- U10 CA180821/CA/NCI NIH HHS/United States
- UG1 CA189858/CA/NCI NIH HHS/United States
- UG1 CA189830/CA/NCI NIH HHS/United States
- UG1 CA239767/CA/NCI NIH HHS/United States
- U10 CA180828/CA/NCI NIH HHS/United States
- UG1 CA233340/CA/NCI NIH HHS/United States
- U10 CA180820/CA/NCI NIH HHS/United States
- U10 CA180888/CA/NCI NIH HHS/United States
- U10 CA180819/CA/NCI NIH HHS/United States
- U10 CA180868/CA/NCI NIH HHS/United States
- U10 CA180826/CA/NCI NIH HHS/United States
- UG1 CA233329/CA/NCI NIH HHS/United States
- UG1 CA189821/CA/NCI NIH HHS/United States
- U10 CA180858/CA/NCI NIH HHS/United States
- U10 CA180846/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

