Pregnancy Alters Innate and Adaptive Immune Responses to Zika Virus Infection in the Reproductive Tract
- PMID: 33127823
- PMCID: PMC7686295
- DOI: 10.4049/jimmunol.2000882
Pregnancy Alters Innate and Adaptive Immune Responses to Zika Virus Infection in the Reproductive Tract
Abstract
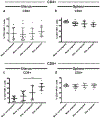
Recent outbreaks of Zika virus (ZIKV) have been associated with birth defects, including microcephaly and neurologic impairment. However, the mechanisms that confer potential susceptibility to ZIKV during pregnancy remain unclear. We hypothesized that poor outcomes from ZIKV infection during pregnancy are due in part to pregnancy-induced alteration of innate immune cell frequencies and cytokine expression. To examine the impact of pregnancy on innate immune responses, we inoculated immunocompetent pregnant and nonpregnant female C57BL/6 mice with 5 × 105 focus-forming units of ZIKV intravaginally. Innate immune cell frequencies and cytokine expression were measured by flow cytometry at day 3 postinfection. Compared with nonpregnant mice, pregnant mice exhibited higher frequencies of uterine macrophages (CD68+) and CD11c+ CD103+ and CD11c+ CD11b+ dendritic cells. Additionally, ZIKV-infected pregnant mice had lower frequencies of CD45+ IL-12+ and CD11b+ IL-12+ cells in the uterus and spleen. Next, we measured the frequencies of Ag-experienced CD4 (CD4+ CD11a+ CD49d+) and CD8 (CD8lo CD11ahi) T cells at day 10 postinfection to determine the impact of pregnancy-associated changes in innate cellular IL-12 responses on the adaptive immune response. We found that pregnant mice had lower frequencies of uterine Ag-experienced CD4 T cells and ZIKV-infected pregnant mice had lower frequencies of uterine Ag-experienced CD8 T cells compared with ZIKV-infected nonpregnant mice. These data show that pregnancy results in altered innate and adaptive immune responses to ZIKV infection in the reproductive tract of mice and that pregnancy-associated immune modulation may play an important role in the severity of acute ZIKV infection.
Copyright © 2020 by The American Association of Immunologists, Inc.
Figures



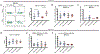



References
-
- Dick GW, Kitchen SF, and Haddow AJ. 1952. Zika virus. I. Isolations and serological specificity. Trans R Soc Trop Med Hyg 46: 509–520. - PubMed
-
- Barjas-Castro ML, Angerami RN, Cunha MS, Suzuki A, Nogueira JS, Rocco IM, Maeda AY, Vasami FG, Katz G, Boin IF, Stucchi RS, Resende MR, Esposito DL, de Souza RP, da Fonseca BA, and Addas-Carvalho M. 2016. Probable transfusion-transmitted Zika virus in Brazil. Transfusion 56: 1684–1688. - PubMed
-
- Meltzer E, Leshem E, Lustig Y, Gottesman G, and Schwartz E. 2016. The Clinical Spectrum of Zika Virus in Returning Travelers. The American journal of medicine 129: 1126–1130. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

