Synthesizing AND gate minigene circuits based on CRISPReader for identification of bladder cancer cells
- PMID: 33127914
- PMCID: PMC7599332
- DOI: 10.1038/s41467-020-19314-7
Synthesizing AND gate minigene circuits based on CRISPReader for identification of bladder cancer cells
Abstract
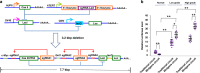
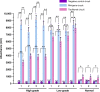
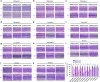
The logical AND gate gene circuit based on the CRISPR-Cas9 system can distinguish bladder cancer cells from normal bladder epithelial cells. However, the layered artificial gene circuits have the problems of high complexity, difficulty in accurately predicting the behavior, and excessive redundancy, which cannot be applied to clinical translation. Here, we construct minigene circuits based on the CRISPReader, a technology used to control promoter-less gene expression in a robust manner. The minigene circuits significantly induce robust gene expression output in bladder cancer cells, but have nearly undetectable gene expression in normal bladder epithelial cells. The minigene circuits show a higher capability for cancer identification and intervention when compared with traditional gene circuits, and could be used for in vivo cancer gene therapy using the all-in-one AAV vector. This approach expands the design ideas and concepts of gene circuits in medical synthetic biology.
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Synthesizing AND gate genetic circuits based on CRISPR-Cas9 for identification of bladder cancer cells.Nat Commun. 2014 Nov 6;5:5393. doi: 10.1038/ncomms6393. Nat Commun. 2014. PMID: 25373919
-
A revolutionary tool: CRISPR technology plays an important role in construction of intelligentized gene circuits.Cell Prolif. 2019 Mar;52(2):e12552. doi: 10.1111/cpr.12552. Epub 2018 Dec 5. Cell Prolif. 2019. PMID: 30520167 Free PMC article. Review.
-
CRISPR-based gene expression control for synthetic gene circuits.Biochem Soc Trans. 2020 Oct 30;48(5):1979-1993. doi: 10.1042/BST20200020. Biochem Soc Trans. 2020. PMID: 32964920 Free PMC article. Review.
-
Multiplexed promoterless gene expression with CRISPReader.Genome Biol. 2019 Jun 3;20(1):113. doi: 10.1186/s13059-019-1712-5. Genome Biol. 2019. PMID: 31159834 Free PMC article.
-
Engineering Synthetic Gene Circuits in Living Cells with CRISPR Technology.Trends Biotechnol. 2016 Jul;34(7):535-547. doi: 10.1016/j.tibtech.2015.12.014. Epub 2016 Jan 22. Trends Biotechnol. 2016. PMID: 26809780 Review.
Cited by
-
A novel CD8+ T cell-related gene signature for predicting the prognosis and immunotherapy efficacy in bladder cancer.Inflamm Res. 2023 Aug;72(8):1665-1687. doi: 10.1007/s00011-023-01772-6. Epub 2023 Aug 14. Inflamm Res. 2023. PMID: 37578544
-
The Progress of the Specific and Rapid Genetic Detection Methods for Ovarian Cancer Diagnosis and Treatment.Technol Cancer Res Treat. 2022 Jan-Dec;21:15330338221114497. doi: 10.1177/15330338221114497. Technol Cancer Res Treat. 2022. PMID: 36062718 Free PMC article. Review.
-
Advancements in genetic circuits as part of intelligent biotherapy for the treatment of bladder cancer: A review.Bladder (San Franc). 2025 Feb 4;12(1):e21200032. doi: 10.14440/bladder.2024.0044. eCollection 2025. Bladder (San Franc). 2025. PMID: 40747118 Free PMC article. Review.
-
Making the Next Generation of Therapeutics: mRNA Meets Synthetic Biology.ACS Synth Biol. 2023 Sep 15;12(9):2505-2515. doi: 10.1021/acssynbio.3c00253. Epub 2023 Sep 6. ACS Synth Biol. 2023. PMID: 37672348 Free PMC article. Review.
-
Advances in Recombinant Adeno-Associated Virus Vectors for Neurodegenerative Diseases.Biomedicines. 2023 Oct 8;11(10):2725. doi: 10.3390/biomedicines11102725. Biomedicines. 2023. PMID: 37893099 Free PMC article. Review.
References
-
- Wrighton KH. Synthetic biology: Multiplex genome engineering in eukaryotes. Nat. Rev. Genet. 2017;19:6. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

