Acute pain and side effects after tramadol in breast cancer patients: results of a prospective double-blind randomized study
- PMID: 33127945
- PMCID: PMC7599328
- DOI: 10.1038/s41598-020-75961-2
Acute pain and side effects after tramadol in breast cancer patients: results of a prospective double-blind randomized study
Abstract
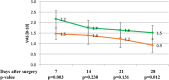
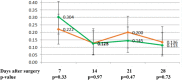
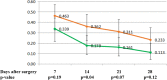
The objective of this study was to evaluate the severity of acute pain and side effects in breast cancer patients postoperatively treated with two regimens of tramadol with paracetamol in a prospective double-blind study. Altogether 117 breast cancer patients who had axillary lymphadenectomy were randomized into two analgesic study groups and the analgesic treatment lasted 4 weeks. Stronger analgesia group received every 8 h 75/650 mg of tramadol with paracetamol, while weaker analgesia group received every 8 h 37.5/325 mg of tramadol with paracetamol. Patients with the higher dose of tramadol had less pain during the 1st and 4th week than patients with the lower dose. Frequency of nausea, vomiting, lymphedema or range of shoulder movement was not significantly different between the two groups of patients. Constipation was significantly more common in the group with stronger analgesia during the 2nd week in comparison to patients with weaker analgesia. The patients who were on 75/650 mg of tramadol with paracetamol had less pain in comparison to patients who were on 37.5/325 mg. Side effects were mild, but common in both groups of patients.
Conflict of interest statement
N. Besic and B. Strazisar have received a research grant Trial KCT 04/2015-doretaonko/si from the pharmaceutical company Krka d.d. Novo mesto. J. Smrekar has received a honorarium for the statistical analysis of our study from the pharmaceutical company Krka d.d. Novo mesto.
Figures
Similar articles
-
Chronic adverse effects after an axillary lymphadenectomy in breast cancer patients after administering weaker and stronger postoperative analgesia: results of a prospective double-blind randomized study.Breast Cancer Res Treat. 2020 Aug;182(3):655-663. doi: 10.1007/s10549-020-05713-3. Epub 2020 Jun 18. Breast Cancer Res Treat. 2020. PMID: 32557338 Clinical Trial.
-
Intravenous paracetamol as adjunctive treatment for postoperative pain after cardiac surgery: a double blind randomized controlled trial.Eur J Cardiothorac Surg. 2007 Sep;32(3):527-31. doi: 10.1016/j.ejcts.2007.05.017. Epub 2007 Jul 23. Eur J Cardiothorac Surg. 2007. PMID: 17643995 Clinical Trial.
-
Tramadol or paracetamol do not effect the diagnostic accuracy of acute abdominal pain with significant pain relief - a prospective, randomized, placebo controlled double blind study.Eur Rev Med Pharmacol Sci. 2012 Dec;16(14):1983-8. Eur Rev Med Pharmacol Sci. 2012. PMID: 23242726 Clinical Trial.
-
[Treating pain after dental surgery: a randomised, controlled, double-blind trial to assess a new formulation of paracetamol, opium powder and caffeine versus tramadol or placebo].Presse Med. 2010 May;39(5):e103-11. doi: 10.1016/j.lpm.2009.09.025. Epub 2010 Jan 18. Presse Med. 2010. PMID: 20080382 Clinical Trial. French.
-
[Tramadol in acute pain].Drugs. 1997;53 Suppl 2:25-33. doi: 10.2165/00003495-199700532-00007. Drugs. 1997. PMID: 9190322 Review. French.
Cited by
-
From Cadaveric Dissection to the Operating Room: A Unilateral Double Intercostobrachial Nerve and the Implications in Axillary Lymph Node Dissection.Cureus. 2023 Mar 24;15(3):e36647. doi: 10.7759/cureus.36647. eCollection 2023 Mar. Cureus. 2023. PMID: 37102027 Free PMC article.
-
Changes in Pain Sensitivity in Treatment for Breast Cancer: A 12-Month Follow-Up Case Series.Int J Environ Res Public Health. 2022 Mar 29;19(7):4055. doi: 10.3390/ijerph19074055. Int J Environ Res Public Health. 2022. PMID: 35409738 Free PMC article.
-
Association of OPRM1, MIR23B, and MIR107 genetic variability with acute pain, chronic pain and adverse effects after postoperative tramadol and paracetamol treatment in breast cancer.Radiol Oncol. 2023 Mar 22;57(1):111-120. doi: 10.2478/raon-2023-0003. eCollection 2023 Mar 1. Radiol Oncol. 2023. PMID: 36942908 Free PMC article. Clinical Trial.
-
Antitumorigenic Effect of Tramadol and Synergistic Effect With Doxorubicin in Human Breast Cancer Cells.Front Oncol. 2022 Jan 26;12:811716. doi: 10.3389/fonc.2022.811716. eCollection 2022. Front Oncol. 2022. PMID: 35155248 Free PMC article.
-
Preparation and identification of anti-breast cancer cells peptides released from yak milk casein.Front Nutr. 2022 Aug 26;9:997514. doi: 10.3389/fnut.2022.997514. eCollection 2022. Front Nutr. 2022. PMID: 36091230 Free PMC article.
References
-
- Gärtner R, et al. Prevalence of and factors associated with persistent pain following breast cancer surgery. JAMA. 2012;18:1985–1992. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

