Risk factors for Coronavirus Disease 2019 (COVID-19) severity and mortality among solid cancer patients and impact of the disease on anticancer treatment: A French nationwide cohort study (GCO-002 CACOVID-19)
- PMID: 33129039
- PMCID: PMC7543792
- DOI: 10.1016/j.ejca.2020.09.035
Risk factors for Coronavirus Disease 2019 (COVID-19) severity and mortality among solid cancer patients and impact of the disease on anticancer treatment: A French nationwide cohort study (GCO-002 CACOVID-19)
Abstract
Background: Cancer patients are thought to have an increased risk of developing severe Coronavirus Disease 2019 (COVID-19) infection and of dying from the disease. In this work, predictive factors for COVID-19 severity and mortality in cancer patients were investigated.
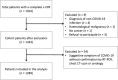
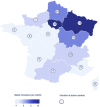
Patients and methods: In this large nationwide retro-prospective cohort study, we collected data on patients with solid tumours and COVID-19 diagnosed between March 1 and 11th June 2020. The primary end-point was all-cause mortality and COVID-19 severity, defined as admission to an intensive care unit (ICU) and/or mechanical ventilation and/or death, was one of the secondary end-points.
Results: From April 4 to 11th June 2020, 1289 patients were analysed. The most frequent cancers were digestive and thoracic. Altogether, 424 (33%) patients had a severe form of COVID-19 and 370 (29%) patients died. In multivariate analysis, independent factors associated with death were male sex (odds ratio 1.73, 95%CI: 1.18-2.52), The Eastern Cooperative Oncology Group Performance Scale (ECOG PS) ≥ 2 (OR 3.23, 95%CI: 2.27-4.61), updated Charlson comorbidity index (OR 1.08, 95%CI: 1.01-1.16) and admission to ICU (OR 3.62, 95%CI 2.14-6.11). The same factors, age along with corticosteroids before COVID-19 diagnosis, and thoracic primary tumour site were independently associated with COVID-19 severity. None of the anticancer treatments administered within the previous 3 months had any effect on mortality or COVID-19 severity, except for cytotoxic chemotherapy in the subgroup of patients with detectable severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) by reverse transcriptase polymerase chain reaction (RT-PCR), which was associated with a slight increase of the risk of death (OR 1.53; 95%CI: 1.00-2.34; p = 0.05). A total of 431 (39%) patients had their systemic anticancer treatment (such as chemotherapy, targeted or immune therapy) interrupted or stopped following diagnosis of COVID-19.
Conclusions: Mortality and COVID-19 severity in cancer patients are high and are associated with general characteristics of patients. We found no deleterious effects of recent anticancer treatments, except for cytotoxic chemotherapy in the RT-PCR-confirmed subgroup of patients. In almost 40% of patients, the systemic anticancer therapy was interrupted or stopped after COVID-19 diagnosis.
Keywords: COVID-19; Cancer; Chemotherapy; Death; Immunotherapy; Intensive care unit; Mechanical ventilation; Mortality; Radiotherapy.
Copyright © 2020 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Conflict of interest statement Pr Lièvre reports grants from BayerLilly, Merck and Novartis, personal fees from AAA, Amgen, Bayer, BMS,Celgene, HalioDx, Incyte, Ipsen, Lilly, Merck, Novartis, Pierre Fabre, Roche, Sandoz, Sanofi, Servier and non-financial support from AAA, Amgen, bayer, Incite, Ipsen, Merck, Novartis, Pierre Fabre, Pfizer, Roche, Sandoz, Servier and Integragen outside of the submitted work. Dr. Turpin reports personal fees from MYLAN, personal fees from MERCK SERONO, personal fees from AMGEN, non-financial support from MERCK-SERONO, non-financial support from SANOFI, non-financial support from PFIZER outside the submitted work. Dr. Ahle reports grants from Biogen, grants from Novartis, grants from Roche, grants from Sanofi, grants from Abbvie, outside the submitted work.Dr Bouché reports personal fees from Merck KGaA, Roche Genentech, Bayer, Astra-Zeneca, Grunenthal, MSD, Amgen, Servier, and Pierre Fabre outside the submitted work. Dr. Neuzillet reports personal fees from SERVIER, other from OSE Immunotherapeutic, grants from ROCHE, personal fees and other from AstraZeneca, personal fees and other from Bristol-Myers Squibb, personal fees from Amgen, personal fees from Merck, personal fees from MSD, personal fees from Novartis, personal fees from Incyte, personal fees from Mylan, personal fees from Baxter, personal fees from Nutricia, personal fees from Fresenius-Kabi, outside the submitted work . Pr Michel reports personal fees from Bayer, Servier, Amgen, Shire and Lilly and non-personal fees from Bayer, Merck, Amgen, Shire, Roche and Lilly outside the submitted work. Dr. Canellas reports personal fees from BMS, personal fees from Aztra Zeneca, non-financial support from Boerhinger Ingheleim, non-financial support from Roche, outside the submitted work . Dr. Wislez reports personal fees from Boeringher Ingelheim, personal fees and non-financial support from ROCHE, personal fees and non-financial support from MSD, personal fees from BMS, personal fees from Astra Zeneca, personal fees from Amgen, outside the submitted work. Dr. Mansi reports personal fees from Roche, personal fees from Eisai, personal fees from Exact Science, personal fees from Novartis, outside the submitted work . Dr Colomba received personnal fees from IPSEN, BMS, Pfizer, Sanofi, GSK outside the submitted work. Pr. Idbaih reports grants and other from Carthera (september 2019), grants from Transgene, grants from Sanofi, grants from Air Liquide, other from Leo Pharma, grants from Nutritheragene, outside the submitted work. Pr. Aparicio reports personal fees and non-financial support from Roche, personal fees from Ipsen, personal fees from Amgen, personal fees from Servier, personal fees from Sanofi, outside the submitted work. All other authors declare no competing interests.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

