Polyethylene glycol 35 ameliorates pancreatic inflammatory response in cerulein-induced acute pancreatitis in rats
- PMID: 33132648
- PMCID: PMC7584060
- DOI: 10.3748/wjg.v26.i39.5970
Polyethylene glycol 35 ameliorates pancreatic inflammatory response in cerulein-induced acute pancreatitis in rats
Abstract
Background: Acute pancreatitis (AP) is a sudden inflammatory process of the pancreas that may also involve surrounding tissues and/or remote organs. Inflammation and parenchymal cell death are common pathological features of this condition and determinants of disease severity. Polyethylene glycols (PEGs) are non-immunogenic, non-toxic water-soluble polymers widely used in biological, chemical, clinical and pharmaceutical settings.
Aim: To evaluate the protective effect of a 35-kDa molecular weight PEG (PEG35) on the pancreatic damage associated to cerulein-induced acute pancreatitis in vivo and in vitro.
Methods: Wistar rats were assigned at random to a control group, a cerulein-induced AP group and a PEG35 treatment group. AP was induced by five hourly intraperitoneal injections of cerulein (50 μg/kg/bw), while the control animals received saline solution. PEG35 was administered intraperitoneally 10 minutes before each cerulein injection in a dose of 10 mg/kg. After AP induction, samples of pancreatic tissue and blood were collected for analysis. AR42J pancreatic acinar cells were treated with increasing concentrations of PEG35 prior to exposure with tumor necrosis factor α (TNFα), staurosporine or cerulein. The severity of AP was determined on the basis of plasma levels of lipase, lactate dehydrogenase activity, pancreatic edema and histological changes. To evaluate the extent of the inflammatory response, the gene expression of inflammation-associated markers was determined in the pancreas and in AR42J-treated cells. Inflammation-induced cell death was also measured in models of in vivo and in vitro pancreatic damage.
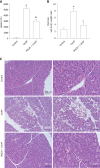
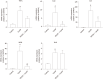
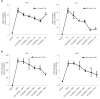
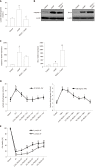
Results: Administration of PEG35 significantly improved pancreatic damage through reduction on lipase levels and tissue edema in cerulein-induced AP rats. The increased associated inflammatory response caused by cerulein administration was attenuated by a decrease in the gene expression of inflammation-related cytokines and inducible nitric oxide synthase enzyme in the pancreas. In contrast, pancreatic tissue mRNA expression of interleukin 10 was markedly increased. PEG35 treatment also protected against inflammation-induced cell death by attenuating lactate dehydrogenase activity and modulating the pancreatic levels of apoptosis regulator protein BCL-2 in cerulein hyperstimulated rats. Furthermore, the activation of pro-inflammatory markers and inflammation-induced cell death in pancreatic acinar cells treated with TNFα, cerulein or staurosporine was significantly reduced by PEG35 treatment, in a dose-dependent manner.
Conclusion: PEG35 ameliorates pancreatic damage in cerulein-induced AP and AR42J-treated cells through the attenuation of the inflammatory response and associated cell death. PEG35 may be a valuable option in the management of AP.
Keywords: AR42J cells; Acute pancreatitis; Cell death; Cytokines; Inflammation; Polyethylene glycols.
©The Author(s) 2020. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: The authors have disclosed that they do not have any conflict of interest.
Figures
Similar articles
-
Polyethylene Glycol 35 (PEG35) Protects against Inflammation in Experimental Acute Necrotizing Pancreatitis and Associated Lung Injury.Int J Mol Sci. 2020 Jan 30;21(3):917. doi: 10.3390/ijms21030917. Int J Mol Sci. 2020. PMID: 32019239 Free PMC article.
-
Karyopherin Alpha 2 Promotes the Inflammatory Response in Rat Pancreatic Acinar Cells Via Facilitating NF-κB Activation.Dig Dis Sci. 2016 Mar;61(3):747-57. doi: 10.1007/s10620-015-3948-6. Epub 2015 Nov 2. Dig Dis Sci. 2016. PMID: 26526450
-
Leptin treatment ameliorates acute lung injury in rats with cerulein-induced acute pancreatitis.World J Gastroenterol. 2007 Jun 7;13(21):2932-8. doi: 10.3748/wjg.v13.i21.2932. World J Gastroenterol. 2007. PMID: 17589942 Free PMC article.
-
Intraarterial Infusion of Lidocaine is Superior to the Subcutaneous Injection of Low Molecular Weight Heparin for Improving the Course of Cerulein-Induced Acute Pancreatitis in Rats.Arch Immunol Ther Exp (Warsz). 2025 Apr 16;73(1). doi: 10.2478/aite-2025-0012. eCollection 2025 Jan 1. Arch Immunol Ther Exp (Warsz). 2025. PMID: 40237149 Review.
-
Arachidonate 15-lipoxygenase: A promising therapeutic target for alleviating inflammation in acute pancreatitis.World J Gastroenterol. 2025 Apr 21;31(15):102752. doi: 10.3748/wjg.v31.i15.102752. World J Gastroenterol. 2025. PMID: 40309226 Free PMC article. Review.
Cited by
-
Enhancing or inhibiting apoptosis? The effects of ucMSC-Ex in the treatment of different degrees of traumatic pancreatitis.Apoptosis. 2022 Aug;27(7-8):521-530. doi: 10.1007/s10495-022-01732-1. Epub 2022 May 25. Apoptosis. 2022. PMID: 35612769
-
Harnessing Polyethylene Glycol 3350 for Enhanced Peripheral Nerve Repair: A Path to Accelerated Recovery.Medicina (Kaunas). 2025 Mar 28;61(4):624. doi: 10.3390/medicina61040624. Medicina (Kaunas). 2025. PMID: 40282915 Free PMC article.
-
Polyethylene Glycol 35 (PEG35) Modulates Exosomal Uptake and Function.Polymers (Basel). 2020 Dec 18;12(12):3044. doi: 10.3390/polym12123044. Polymers (Basel). 2020. PMID: 33353210 Free PMC article.
-
Impact of Hypothermic Perfusion on Immune Responses and Sterile Inflammation in a Preclinical Model of Pancreatic Transplantation.Transplant Direct. 2024 Dec 13;11(1):e1743. doi: 10.1097/TXD.0000000000001743. eCollection 2025 Jan. Transplant Direct. 2024. PMID: 39687511 Free PMC article.
References
-
- Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, Tsiotos GG, Vege SS Acute Pancreatitis Classification Working Group. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62:102–111. - PubMed
-
- van Santvoort HC, Bakker OJ, Bollen TL, Besselink MG, Ahmed Ali U, Schrijver AM, Boermeester MA, van Goor H, Dejong CH, van Eijck CH, van Ramshorst B, Schaapherder AF, van der Harst E, Hofker S, Nieuwenhuijs VB, Brink MA, Kruyt PM, Manusama ER, van der Schelling GP, Karsten T, Hesselink EJ, van Laarhoven CJ, Rosman C, Bosscha K, de Wit RJ, Houdijk AP, Cuesta MA, Wahab PJ, Gooszen HG Dutch Pancreatitis Study Group. A conservative and minimally invasive approach to necrotizing pancreatitis improves outcome. Gastroenterology. 2011;141:1254–1263. - PubMed
-
- Sendler M, van den Brandt C, Glaubitz J, Wilden A, Golchert J, Weiss FU, Homuth G, De Freitas Chama LL, Mishra N, Mahajan UM, Bossaller L, Völker U, Bröker BM, Mayerle J, Lerch MM. NLRP3 Inflammasome Regulates Development of Systemic Inflammatory Response and Compensatory Anti-Inflammatory Response Syndromes in Mice With Acute Pancreatitis. Gastroenterology. 2020;158:253–269.e14. - PubMed

