Early Auditory Experience Modifies Neuronal Firing Properties in the Zebra Finch Auditory Cortex
- PMID: 33132855
- PMCID: PMC7578418
- DOI: 10.3389/fncir.2020.570174
Early Auditory Experience Modifies Neuronal Firing Properties in the Zebra Finch Auditory Cortex
Abstract
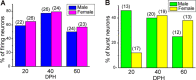
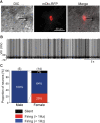
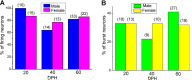
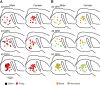
Songbirds learn to sing much as humans learn to speak. In zebra finches, one of the premier songbird models, males learn to sing for later courtship through a multistep learning process during the developmental period. They first listen to and memorize the song of a tutor (normally their father) during the sensory learning period. Then, in the subsequent sensory-motor learning phase (with large overlap), they match their vocalizations to the memorized tutor song via auditory feedback and develop their own unique songs, which they maintain throughout their lives. Previous studies have suggested that memories of tutor songs are shaped in the caudomedial nidopallium (NCM) of the brain, which is analogous to the mammalian higher auditory cortex. Isolation during development, which extends the sensory learning period in males, alters song preference in adult females, and NCM inactivation decreases song preference. However, the development of neurophysiological properties of neurons in this area and the effect of isolation on these neurons have not yet been explained. Here, we performed whole-cell patch-clamp recording on NCM neurons from juvenile zebra finches during the sensory learning period, 20, 40, or 60 days post-hatching (DPH) and examined their neurophysiological properties. In contrast to previous reports in adult NCM neurons, the majority of NCM neurons of juvenile zebra finches showed spontaneous firing with or without burst firing patterns, and the percentage of neurons that fired increased in the middle of the sensory learning period (40 DPH) and then decreased at the end (60 DPH) in both males and females. We further found that auditory isolation from tutor songs alters developmental changes in the proportions of firing neurons both in males and females, and also changes those of burst neurons differently between males that sing and females that do not. Taken together, these findings suggest that NCM neurons develop their neurophysiological properties depending on auditory experiences during the sensory song learning period, which underlies memory formation for song learning in males and song discrimination in females.
Keywords: auditory cortex; auditory experience; critical period; firing properties; song learning; songbird; zebra finch.
Copyright © 2020 Kudo, Morohashi and Yazaki-Sugiyama.
Figures


Similar articles
-
Auditory experience-dependent cortical circuit shaping for memory formation in bird song learning.Nat Commun. 2016 Jun 21;7:11946. doi: 10.1038/ncomms11946. Nat Commun. 2016. PMID: 27327620 Free PMC article.
-
Experience- and Sex-Dependent Intrinsic Plasticity in the Zebra Finch Auditory Cortex during Song Memorization.J Neurosci. 2020 Mar 4;40(10):2047-2055. doi: 10.1523/JNEUROSCI.2137-19.2019. Epub 2020 Jan 14. J Neurosci. 2020. PMID: 31937558 Free PMC article.
-
Development of neural responsivity to vocal sounds in higher level auditory cortex of songbirds.J Neurophysiol. 2014 Jul 1;112(1):81-94. doi: 10.1152/jn.00484.2013. Epub 2014 Apr 2. J Neurophysiol. 2014. PMID: 24694936 Free PMC article.
-
The sensitive period for auditory-vocal learning in the zebra finch: Consequences of limited-model availability and multiple-tutor paradigms on song imitation.Behav Processes. 2019 Jun;163:5-12. doi: 10.1016/j.beproc.2017.07.007. Epub 2017 Jul 23. Behav Processes. 2019. PMID: 28743517 Free PMC article. Review.
-
Birdsong memory and the brain: in search of the template.Neurosci Biobehav Rev. 2015 Mar;50:41-55. doi: 10.1016/j.neubiorev.2014.11.019. Epub 2014 Nov 29. Neurosci Biobehav Rev. 2015. PMID: 25459663 Review.
Cited by
-
Adult-like neural representation of species-specific songs in the auditory forebrain of zebra finch nestlings.Dev Neurobiol. 2021 Mar;81(2):123-138. doi: 10.1002/dneu.22802. Epub 2021 Jan 29. Dev Neurobiol. 2021. PMID: 33369121 Free PMC article.
-
Lateralized local circuit tuning in female mouse auditory cortex.Neurosci Res. 2025 Jul;216:104897. doi: 10.1016/j.neures.2025.03.009. Epub 2025 Apr 4. Neurosci Res. 2025. PMID: 40189152 Free PMC article.
-
The songbird connectome (OSCINE-NET.ORG): structure-function organization beyond the canonical vocal control network.BMC Neurosci. 2024 Dec 27;25(1):79. doi: 10.1186/s12868-024-00919-3. BMC Neurosci. 2024. PMID: 39731002 Free PMC article.
-
Neuroestrogens rapidly shape auditory circuits to support communication learning and perception: Evidence from songbirds.Horm Behav. 2018 Aug;104:77-87. doi: 10.1016/j.yhbeh.2018.03.007. Epub 2018 Mar 30. Horm Behav. 2018. PMID: 29555375 Free PMC article. Review.
-
Social and auditory experience shapes forebrain responsiveness in zebra finches before the sensitive period of vocal learning.J Exp Biol. 2024 Nov 1;227(21):jeb247956. doi: 10.1242/jeb.247956. Epub 2024 Oct 25. J Exp Biol. 2024. PMID: 39263850 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

