Hypervalent Iodine Reagents in Palladium-Catalyzed Oxidative Cross-Coupling Reactions
- PMID: 33134246
- PMCID: PMC7553084
- DOI: 10.3389/fchem.2020.00705
Hypervalent Iodine Reagents in Palladium-Catalyzed Oxidative Cross-Coupling Reactions
Abstract
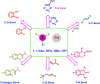
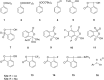
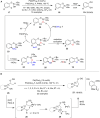
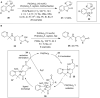
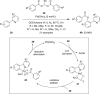
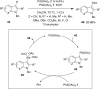
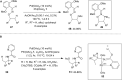
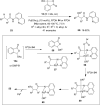
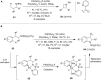
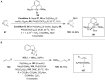
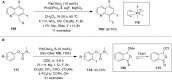
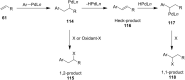
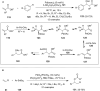
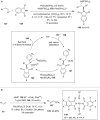
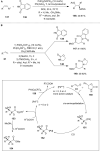
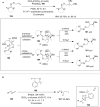
Hypervalent iodine compounds are valuable and versatile reagents in synthetic organic chemistry, generating a diverse array of useful organic molecules. Owing to their non-toxic and environmentally friendly features, these reagents find potential applications in various oxidative functionalization reactions. In recent years, the use of hypervalent iodine reagents in palladium-catalyzed transformations has been widely studied as they are strong electrophiles and powerful oxidizing agents. For instance, extensive work has been carried out in the field of C-H bond functionalization via Pd-catalysis using hypervalent iodine reagents as oxidants. In addition, nowadays, iodine(III) reagents have been frequently employed as arylating agents in Pd-catalyzed C-H arylation or Heck-type cross-coupling reactions. In this review, recent advancements in the area of palladium-catalyzed oxidative cross-coupling reactions using hypervalent iodine reagents are summarized in detail.
Keywords: bond formation; catalyst; hypervalent iodine reagents; oxidant; palladium.
Copyright © 2020 Shetgaonkar and Singh.
Figures


Similar articles
-
Palladium-Catalyzed Organic Reactions Involving Hypervalent Iodine Reagents.Molecules. 2022 Jun 17;27(12):3900. doi: 10.3390/molecules27123900. Molecules. 2022. PMID: 35745020 Free PMC article. Review.
-
Non-Palladium-Catalyzed Oxidative Coupling Reactions Using Hypervalent Iodine Reagents.Front Chem. 2022 Jul 1;10:909250. doi: 10.3389/fchem.2022.909250. eCollection 2022. Front Chem. 2022. PMID: 35844643 Free PMC article. Review.
-
Reactions of hypervalent iodine reagents with palladium: mechanisms and applications in organic synthesis.Inorg Chem. 2007 Mar 19;46(6):1924-35. doi: 10.1021/ic0620337. Inorg Chem. 2007. PMID: 17348723
-
Hypervalent iodine-catalyzed oxidative functionalizations including stereoselective reactions.Chem Asian J. 2014 Apr;9(4):950-71. doi: 10.1002/asia.201301582. Epub 2014 Feb 12. Chem Asian J. 2014. PMID: 24523252
-
Hypervalent-Iodine(III)-Mediated Oxidative Methodology for the Synthesis of Fused Triazoles.Chem Asian J. 2016 Jul 20;11(14):1988-2000. doi: 10.1002/asia.201600119. Epub 2016 Jun 6. Chem Asian J. 2016. PMID: 27123538 Review.
Cited by
-
Iodine(V)-Based Oxidants in Oxidation Reactions.Molecules. 2023 Jul 6;28(13):5250. doi: 10.3390/molecules28135250. Molecules. 2023. PMID: 37446912 Free PMC article. Review.
-
Palladium-Catalyzed Organic Reactions Involving Hypervalent Iodine Reagents.Molecules. 2022 Jun 17;27(12):3900. doi: 10.3390/molecules27123900. Molecules. 2022. PMID: 35745020 Free PMC article. Review.
-
Non-Palladium-Catalyzed Oxidative Coupling Reactions Using Hypervalent Iodine Reagents.Front Chem. 2022 Jul 1;10:909250. doi: 10.3389/fchem.2022.909250. eCollection 2022. Front Chem. 2022. PMID: 35844643 Free PMC article. Review.
-
Iodine(III)-Containing Reagents in Photo-Assisted and Photo-Catalyzed Organic Synthesis.Molecules. 2025 Feb 8;30(4):784. doi: 10.3390/molecules30040784. Molecules. 2025. PMID: 40005096 Free PMC article. Review.
-
Metal Centers as Nucleophiles: Oxymoron of Halogen Bond-Involving Crystal Engineering.Chemistry. 2022 Jan 10;28(2):e202103173. doi: 10.1002/chem.202103173. Epub 2021 Oct 29. Chemistry. 2022. PMID: 34623005 Free PMC article. Review.
References
Publication types
LinkOut - more resources
Full Text Sources

