A spider in motion: facets of sensory guidance
- PMID: 33135112
- PMCID: PMC8046691
- DOI: 10.1007/s00359-020-01449-z
A spider in motion: facets of sensory guidance
Abstract
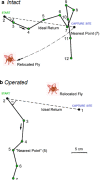
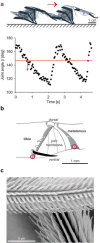
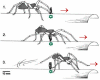
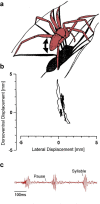
Spiders show a broad range of motions in addition to walking and running with their eight coordinated legs taking them towards their resources and away from danger. The usefulness of all these motions depends on the ability to control and adjust them to changing environmental conditions. A remarkable wealth of sensory receptors guarantees the necessary guidance. Many facets of such guidance have emerged from neuroethological research on the wandering spider Cupiennius salei and its allies, although sensori-motor control was not the main focus of this work. The present review may serve as a springboard for future studies aiming towards a more complete understanding of the spider's control of its different types of motion. Among the topics shortly addressed are the involvement of lyriform slit sensilla in path integration, muscle reflexes in the walking legs, the monitoring of joint movement, the neuromuscular control of body raising, the generation of vibratory courtship signals, the sensory guidance of the jump to flying prey and the triggering of spiderling dispersal behavior. Finally, the interaction of sensors on different legs in oriented turning behavior and that of the sensory systems for substrate vibration and medium flow are addressed.
Keywords: Mechanoreception; Neuroethology; Sensory control; Sensory ecology; Spider motion.
Conflict of interest statement
The author declares no conflict of interest.
Figures


Similar articles
-
In search of differences between the two types of sensory cells innervating spider slit sensilla (Cupiennius salei Keys.).J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2009 Nov;195(11):1031-41. doi: 10.1007/s00359-009-0477-9. Epub 2009 Sep 17. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2009. PMID: 19760269
-
Micromechanical properties of strain-sensitive lyriform organs of a wandering spider (Cupiennius salei).Acta Biomater. 2016 Sep 1;41:40-51. doi: 10.1016/j.actbio.2016.06.009. Epub 2016 Jun 6. Acta Biomater. 2016. PMID: 27282647
-
Spider joint hair sensilla: adaptation to proprioreceptive stimulation.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2015 Feb;201(2):235-48. doi: 10.1007/s00359-014-0965-4. Epub 2014 Nov 15. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2015. PMID: 25398577
-
From stress and strain to spikes: mechanotransduction in spider slit sensilla.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2002 Nov;188(10):739-52. doi: 10.1007/s00359-002-0363-1. Epub 2002 Oct 31. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2002. PMID: 12466950 Review.
-
Mechanotransduction in spider slit sensilla.Can J Physiol Pharmacol. 2004 Aug-Sep;82(8-9):541-8. doi: 10.1139/y04-031. Can J Physiol Pharmacol. 2004. PMID: 15523511 Review.
Cited by
-
Moving in an Uncertain World: Robust and Adaptive Control of Locomotion from Organisms to Machine Intelligence.Integr Comp Biol. 2024 Nov 21;64(5):1390-1407. doi: 10.1093/icb/icae121. Integr Comp Biol. 2024. PMID: 39090982 Free PMC article. Review.
-
Locomotion and kinematics of arachnids.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2021 Mar;207(2):99-103. doi: 10.1007/s00359-021-01478-2. Epub 2021 Mar 18. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2021. PMID: 33738532 Free PMC article.
-
Measuring strain in the exoskeleton of spiders-virtues and caveats.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2021 Mar;207(2):191-204. doi: 10.1007/s00359-020-01458-y. Epub 2021 Jan 18. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2021. PMID: 33459819 Free PMC article. Review.
-
Aerodynamics and the role of the earth's electric field in the spiders' ballooning flight.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2021 Mar;207(2):219-236. doi: 10.1007/s00359-021-01474-6. Epub 2021 Mar 13. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2021. PMID: 33712884 Review.
-
Spider dynamics under vertical vibration and its implications for biological vibration sensing.J R Soc Interface. 2023 Sep;20(206):20230365. doi: 10.1098/rsif.2023.0365. Epub 2023 Sep 13. J R Soc Interface. 2023. PMID: 37700709 Free PMC article.
References
-
- Anton S, Barth FG. Central nervous projection patterns of trichobothria and other cuticular sensilla in the wandering spider Cupiennius salei (Arachnida, Araneae) Zoomorphol. 1993;113:21–32.
-
- Babu KS, Barth FG. Neuroanatomy of the central nervous system of a wandering spider, Cupiennius salei (Arachnida, Araneae) Zoomorphol. 1984;104:344–359.
-
- Babu KS, Barth FG. Central nervous projections of mechanoreceptors in the spider Cupiennius salei KEYS. Cell Tissue Res. 1989;258:69–82.
-
- Barnes WJP, Barth FG (1991) Sensory control of locomotor mode in semi-aquatic spiders. In: Armstrong DM, Bush BMH (eds) Locomotor neural mechanisms in arthropods and vertebrates, chap 10. Manchester Univ Press, Manchester, pp 105–116
-
- Barth FG (1997) Vibratory communication in spiders: Adaptation and compromise at many levels. In: Lehrer M (ed) Orientation and communication in arthropods. Birkhäuser, pp.247–272
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

