Short-term ex-vivo exposure to hydrogen sulfide enhances murine hematopoietic stem and progenitor cell migration, homing, and proliferation
- PMID: 33135550
- PMCID: PMC7671055
- DOI: 10.1080/19336918.2020.1842131
Short-term ex-vivo exposure to hydrogen sulfide enhances murine hematopoietic stem and progenitor cell migration, homing, and proliferation
Abstract
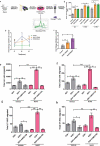
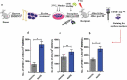
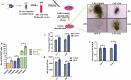
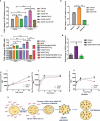
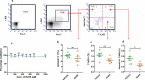
For successful transplantation of Hematopoietic Stem cells (HSCs), it is quite necessary that efficient homing, engraftment and retention of HSC self-renewal capacity takes place, which is often restricted due to inadequate number of adult HSCs. Here, we report that short-term ex-vivo treatment of mouse bone marrow mononuclear cells (BMMNCs) to Sodium Hydrogen Sulfide (NaHS, hydrogen sulfide-H2S donor) can be used as a possible strategy to overcome such hurdle. H2S increases the expression of CXCR4 on HSPCs, enhancing their migration toward SDF-1α in-vitro and thus homing to BM niche. . Additionally, in-vitro studies revealed that H2S has a role in activating mitochondria, thus, pushing quiescent HSCs into division. These results suggest a readily available and cost-effective method to facilitate efficient HSC transplantation.
Keywords: CXCR4 expression; Stem cell migration; bone marrow transplantation; homing; mitochondrial function.
Conflict of interest statement
No potential conflict of interest was reported by the authors.
Figures

Similar articles
-
Prostaglandin E2 enhances hematopoietic stem cell homing, survival, and proliferation.Blood. 2009 May 28;113(22):5444-55. doi: 10.1182/blood-2009-01-201335. Epub 2009 Mar 26. Blood. 2009. PMID: 19324903 Free PMC article.
-
Enhanced functional response to CXCL12/SDF-1 through retroviral overexpression of CXCR4 on M07e cells: implications for hematopoietic stem cell transplantation.Stem Cells Dev. 2006 Jun;15(3):325-33. doi: 10.1089/scd.2006.15.325. Stem Cells Dev. 2006. PMID: 16846371
-
Dimethyl Sulfoxide (DMSO) Increases Percentage of CXCR4(+) Hematopoietic Stem/Progenitor Cells, Their Responsiveness to an SDF-1 Gradient, Homing Capacities, and Survival.Cell Transplant. 2016;25(7):1247-57. doi: 10.3727/096368915X689424. Epub 2015 Sep 4. Cell Transplant. 2016. PMID: 26345294
-
Emerging Strategies to Enhance Homing and Engraftment of Hematopoietic Stem Cells.Stem Cell Rev Rep. 2016 Feb;12(1):121-8. doi: 10.1007/s12015-015-9625-5. Stem Cell Rev Rep. 2016. PMID: 26400757 Free PMC article. Review.
-
Stromal-derived factor-1/CXCR4 signaling: indispensable role in homing and engraftment of hematopoietic stem cells in bone marrow.Stem Cells Dev. 2011 Jun;20(6):933-46. doi: 10.1089/scd.2010.0263. Epub 2011 Feb 2. Stem Cells Dev. 2011. PMID: 21186999 Review.
Cited by
-
Targeting the redox system for cardiovascular regeneration in aging.Aging Cell. 2023 Dec;22(12):e14020. doi: 10.1111/acel.14020. Epub 2023 Nov 13. Aging Cell. 2023. PMID: 37957823 Free PMC article. Review.
-
Emerging roles of cystathionine β-synthase in various forms of cancer.Redox Biol. 2022 Jul;53:102331. doi: 10.1016/j.redox.2022.102331. Epub 2022 May 10. Redox Biol. 2022. PMID: 35618601 Free PMC article. Review.
References
-
- Hu X, Yu SP, Fraser JL, et al. Transplantation of hypoxia-preconditioned mesenchymal stem cells improves infarcted heart function via enhanced survival of implanted cells and angiogenesis. Cardiopulm Support Physiol. 2008;135(4):799–808. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
