Novel Pathogenic Sequence Variants in NR2E3 and Clinical Findings in Three Patients
- PMID: 33138239
- PMCID: PMC7716234
- DOI: 10.3390/genes11111288
Novel Pathogenic Sequence Variants in NR2E3 and Clinical Findings in Three Patients
Abstract
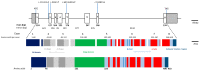
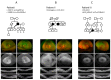
A retrospective review of the clinical records of patients seen at the Oxford Eye Hospital identified as having NR2E3 mutations was performed. The data included symptoms, best-corrected visual acuity, multimodal retinal imaging, visual fields and electrophysiology testing. Three participants were identified with biallelic NR2E3 pathogenic sequence variants detected using a targeted NGS gene panel, two of which were novel. Participant I was a Nepalese male aged 68 years, and participants II and III were white Caucasian females aged 69 and 10 years old, respectively. All three had childhood onset nyctalopia, a progressive decrease in central vision, and visual field loss. Patients I and III had photopsia, patient II had photosensitivity and patient III also had photophobia. Visual acuities in patients I and II were preserved even into the seventh decade, with the worst visual acuity measured at 6/36. Visual field constriction was severe in participant I, less so in II, and fields were full to bright targets targets in participant III. Electrophysiology testing in all three demonstrated loss of rod function. The three patients share some of the typical distinctive features of NR2E3 retinopathies, as well as a novel clinical observation of foveal ellipsoid thickening.
Keywords: Goldmann–Favre syndrome; NR2E3; autosomal recessive and autosomal dominant retinitis pigmentosa; ellipsoid zone; enhanced S-cone syndrome; inherited retinal degeneration; pigmentary clumping; retinal dystrophy.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

References
-
- Haider N.B., Jacobson S.G., Cideciyan A.V., Swiderski R., Streb L.M., Searby C., Beck G., Hockey R., Hanna D.B., Gorman S., et al. Mutation of a nuclear receptor gene, NR2E3, causes enhanced S cone syndrome, a disorder of retinal cell fate. Nat. Genet. 2000;24:127–131. doi: 10.1038/72777. - DOI - PubMed
-
- Milam A.H., Rose L., Cideciyan A.V., Barakat M.R., Tang W.X., Gupta N., Aleman T.S., Wright A.F., Stone E.M., Sheffield V.C., et al. The nuclear receptor NR2E3 plays a role in human retinal photoreceptor differentiation and degeneration. Proc. Natl. Acad. Sci. USA. 2002;99:473–478. doi: 10.1073/pnas.022533099. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

